Ecological effects of extreme climatic events on riverine ecosystems: insights from Australia
Transcript of Ecological effects of extreme climatic events on riverine ecosystems: insights from Australia
Ecological effects of extreme climatic events on riverineecosystems: insights from Australia
CATHERINE LEIGH*, ALEX BUSH† , EVAN T. HARRISON‡ , SUSIE S. HO§, LAURISSE LUKE¶,
ROBERT J. ROLLS¶ AND MARK E. LEDGER**
*Irstea, UR MALY, Villeurbanne, France†Department of Biological Sciences, Macquarie University, North Ryde, NSW, Australia‡Institute for Applied Ecology, University of Canberra, Bruce, ACT, Australia§School of Biological Sciences, Monash University, Clayton, Vic., Australia¶Australian Rivers Institute, Griffith University, Nathan, Qld, Australia
**School of Geography, Earth and Environmental Sciences, University of Birmingham, Edgbaston, Birmingham, U.K.
SUMMARY
1. Climate extremes and their physical impacts – including droughts, fires, floods, heat waves, storm
surges and tropical cyclones – are important structuring forces in riverine ecosystems. Climate
change is expected to increase the future occurrence of extremes, with potentially devastating effects
on rivers and streams. We synthesise knowledge of extremes and their impacts on riverine ecosys-
tems in Australia, a country for which projected changes in event characteristics reflect global trends.
2. Hydrologic extremes play a major structuring role in river ecology across Australia. Droughts alter
water quality and reduce habitat availability, driving organisms to refugia. Extreme floods increase
hydrological connectivity and trigger booms in productivity, but can also alter channel morphology
and cause disturbances such as hypoxic blackwater events.
3. Tropical cyclones and post-cyclonic floods damage riparian vegetation, erode stream banks and
alter water quality. Cyclone-induced delivery of large woody debris provides important instream
habitat, although the wider ecological consequences of tropical cyclones are uncertain.
4. Wildfires destroy catchment vegetation and expose soils, increasing inputs of fine sediment and
nutrients to streams, particularly when followed by heavy rains.
5. Research on the impacts of heat waves and storm surges is scarce, but data on temperature and
salinity tolerances, respectively, may provide some insight into ecological responses.
6. We identify research gaps and hypotheses to guide future research on the ecology of extreme
climate events in Australia and beyond. A range of phenomenological, experimental and modelling
approaches is needed to develop a mechanistic understanding of the ecological impact of extreme
events and inform prediction of responses to future change.
Keywords: biota, climate change, climate extremes, ecosystem functioning, river
Introduction
The frequency and intensity of extreme climatic events is
increasing (IPCC, 2012). Extreme events are statistically
rare or unusual weather or climatic occurrences, such as
extremes of precipitation or temperature, which can have
severe natural impacts on the environment, including
major floods, hydrologic droughts and fire (IPCC, 2012).
Shifts in the regimes and coincidence of extreme events
could alter the biodiversity and ecosystem functioning of
rivers (Paine, Tegner & Johnson, 1998; Jones, 2013).
Understanding the ecological effects of these events is
essential to predict ecological responses to future change.
In this study, we synthesise the knowledge of extremes
and their impact on riverine ecosystems in Australia, a
continent that has experienced multiple record-breaking
events in the 21st century (Steffen, Hughes & Karoly,
2013).
Correspondence: Catherine Leigh, Irstea, UR MALY, 5 rue de la Doua, CS70077, 69626 Villeurbanne Cedex, France.
E-mail: [email protected]
© 2014 John Wiley & Sons Ltd 1
Freshwater Biology (2014) doi:10.1111/fwb.12515
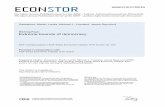
Australia spans multiple climate zones (Fig. 1a–c) and
its rivers have a correspondingly wide range of flow
regimes, from predictably perennial to unpredictably
intermittent (Kennard et al., 2010). Although many spe-
cies in these systems are adapted to natural variability
in climate and river flow, the persistence of taxa, partic-
ularly those with restricted geographic and climatic
ranges, may be threatened by climate extremes (Hughes,
2011; Morrongiello et al., 2011). As the observed and
projected occurrence of extreme events in Australia
reflects that in many other regions of the world (IPCC,
2012), ecological responses in Australia can help under-
stand events elsewhere.
We focus on six types of extreme events identified by
the Australian Climate Commission as a threat to Aus-
tralian ecosystems (Steffen et al., 2013): heat waves and
hot days, fires, droughts, heavy rainfall and floods, trop-
ical cyclones, and storm surges and coastal flooding. We
examine their effects as reported in the literature, iden-
tify key knowledge gaps and propose hypotheses to
stimulate research.
Heat waves and hot days
Definitions, observations and projections
Heat waves, sometimes referred to as warm spells, are
periods of abnormally hot weather, but more precise
definitions vary regionally (IPCC, 2012; Perkins &
Alexander, 2013). In Australia, heat waves are defined
as at least three consecutive days of high maximum and
minimum temperatures that are unusual for a location
(BOM, 2014a). High temperature extremes also include
hot and very hot days (>35 °C and >40 °C, respectively)
and warm days and nights (temperatures > 90th percen-
tile for any 5-day window across the annual cycle)
(BOM, 2014b). The duration and frequency of heat
waves have increased in Australia since the 1970s, and
the hottest days of heat waves have become hotter (Per-
kins & Alexander, 2013). The warmest year on record
(2013) included a national summer heat wave c. 3 weeks
long (BOM, 2013). Climate projections indicate that the
duration of heat waves and the number of hot days will
continue to increase across Australia, consistent with
projected global trends (Alexander & Arblaster, 2009;
IPCC, 2012).
Ecological effects
The ecological effects of heat waves and hot days are
less well understood than those of hydrologic extremes.
However, long-term research on the European summer
heat wave of 2003 indicated these events can have
severe, lasting impacts on benthic assemblages (Mou-
thon & Daufresne, 2006). In Australia, the prospect of
unprecedented thermal extremes has renewed interest in
upper thermal limits (UTLs) of aquatic biota (e.g. Stew-
art et al., 2013a). UTLs are typically determined experi-
mentally using dynamic, non-lethal, behaviour-based
methods (e.g. critical thermal method) or static, lethal
methods (e.g. lethal temperature), with the former gen-
erally considered the better approximation to field con-
ditions. UTLs and the vulnerability of biota to heat
waves and hot days are likely to depend on a variety of
factors, including antecedent ambient temperature and
the rate and duration of warming in the environment,
but data are scarce (Beitinger, Bennett & McCauley,
2000; Dallas & Rivers-Moore, 2012).
Air temperatures in Australia regularly exceed the
UTLs of many aquatic taxa during the warmest months
of the year, although the periods of exceedance may
only span a few hours in a daily cycle (Davies et al.,
2004), and this may also be the case during heat waves.
Fully aquatic species and those with long aquatic life
stages may be less vulnerable to extreme air tempera-
tures than semi-aquatic species or those with extended
aerial life stages, particularly where water temperatures
remain cool, or at least below UTLs, due to evaporative
cooling (Bogan et al., 2006), riparian shading or ground-
water inputs. However, water temperatures that
approach or exceed UTLs during heat waves, particu-
larly those of extended duration or extreme intensity,
and in hydrologically disconnected, shallow or still
waters, could eliminate sensitive taxa (e.g. Matthews,
Surat & Hill, 1982). The projected intensification of heat
waves threatens the survival of many riverine species
(Fig. 2), and persistence may depend on adaptive man-
agement such as riparian revegetation and provision of
cool-water refuges (Robson et al., 2013).
Research to determine UTLs for stream invertebrates
has revealed marked contrasts in vulnerability to
extremes both within and among taxonomic groups. For
instance, UTLs of Ephemeroptera and Plecoptera
nymphs are generally lower than those of Odonata and
Coleoptera larvae (e.g. Stewart et al., 2013a), although
the mean UTL for each order varies substantially among
geographic regions (e.g. southern versus northern taxa;
Davies et al., 2004). Some Ephemeroptera from south-
east Queensland can survive water temperatures >30 °C
for at least 72 h (L. Luke, unpubl. data), which is well
above the global mean UTL for this group (c. 22 °C;
Davies et al., 2004; Stewart et al., 2013a).
© 2014 John Wiley & Sons Ltd, Freshwater Biology, doi: 10.1111/fwb.12515
2 C. Leigh et al.
Temperate, Mediterranean and Subalpine
GrasslandDesert
Tropical and Equatorial
SubtropicalArid and Semi-arid
(e)
(f)
Darwin
Cairns
PerthSydney
Brisbane
Canberra,
Hobart,
Melbourne
Adelaide
Alice Springs
(d)(a)
NT
QLD
VIC
WA
TAS
SANSW
(g)
Annu
al ra
infa
ll (m
m)
Annu
al m
ean
tem
pera
ture
(°C
)
(b)
(c)
0
500
1000
1500
2000
2500
Dar
win
Bris
bane
Syd
ney
Mel
bour
ne
Hob
art
Ade
laid
e
Per
th
Alic
e Sp
rings
05
101520253035
Dar
win
Bris
bane
Syd
ney
Mel
bour
ne
Hob
art
Ade
laid
e
Per
th
Alic
e Sp
rings
5th percen�le
95th percen�leMedian
Daily minimaDaily maxima
ACT
© 2014 John Wiley & Sons Ltd, Freshwater Biology, doi: 10.1111/fwb.12515
Extreme events and riverine ecosystems 3
Vulnerability to heat waves can be influenced by
behavioural or physiological responses to warming.
For example, high temperatures may influence the
dispersal of some species (e.g. Hassall & Thompson,
2008), limiting escape of intolerable conditions. Sublethal
effects could also alter sex ratios in surviving populations.
For instance, temperature extremes forecast for the year
2100 simulated in a mesocosm experiment increased
emergence rates of male Ulmerophlebia pipinna mayflies
more so than females, implying a change in the sex ratio
that could have demographic and fitness consequences
and cause local extinctions (Thompson et al., 2013a).
Knowledge gaps
Knowledge of thermal tolerance in aquatic organisms is
limited to coarse levels of taxonomic resolution, and less
is known about thermal tolerance among closely related
species or across populations (Chessman, 2009, 2013;
Stewart et al., 2013a). Organismal responses to extreme
temperature may be context dependent (Caissie, 2006;
Verberk & Bilton, 2013), varying among habitats, and
contingent upon the modifying influence of other
extreme events or stressors. For instance, thermal
impacts on biota may be exacerbated during drought
when instream temperatures are less buffered against
temperature changes in air (Davies et al., 2004; Fig. 2),
but data are scarce. More research is needed to identify
heat wave and hot day refugia for the most sensitive
taxa to direct management efforts, and there is a critical
absence of research specifically examining effects of heat
waves and hot days at the community and ecosystem
levels.
Droughts
Definitions, observations and projections
Hydrological drought is a period of below-typical flow
that is unusual in its duration, extent or intensity
(Humphries & Baldwin, 2003). Drought is seasonal in
many systems, but here, we focus solely on ‘supra-sea-
sonal’ events: unpredictable droughts extending beyond
seasonal periods, sometimes lasting >10 years (‘mega-
droughts’) (Lake, 2003; IPCC, 2012). Drought is a key
element of climatic and hydrological variability in Aus-
tralia. The Millennium Drought in south-eastern Austra-
lia was one of the most severe hydrological and
meteorological mega-droughts on record, lasting
between 1997 and 2009 (Gallant & Gergis, 2011; Timbal
& Fawcett, 2012; Fig. 1g). Mega-drought has also
affected the south-western region of Western Australia,
which has been in drought since the mid-1970s, with
rainfall 10–20% below long-term averages (NCC, 2010).
Climate projections indicate southern Australia will
Fig. 1 Australia has a range of climate zones (a), with regional variation in spatiotemporal patterns of temperature and rainfall (b, c).
Examples of extreme events include Cyclone Larry in March 2006 (d: aerial image of Upper North Johnstone River, post-event, showing
treefall and leaf denudation in the riparian zone; credit S. Turton), flood across much of south-east QLD in January 2011 (e: Gregors Creek,
post-event, during which flood waters reached the mature treeline above river banks; credit L. Luke), the 2003 wildfires in the ACT
(f: aerial image of Cotter River, post-event, showing burned vegetation across the catchment; credit T. Nelson), and the Millennium Drought
(g: a drying waterbody in the Ovens River catchment, 2009; credit S.S. Ho). Climate-zone mapping data and summary statistics sourced
from www.bom.gov.au, with statistics calculated for the reference period 1 January 1961 to 31 December 1990. ACT, Australian Capital
Territory; NSW, New South Wales; NT, Northern Territory; QLD, Queensland; SA, South Australia; TAS, Tasmania; VIC, Victoria; WA,
Western Australia.
Heat waves & hot days
Δ Emergence rates of aqua�c
macroinvertebrates
Disrup�on or ins�ga�on of spawning or
dispersal cues in some biota
Δ Aqua�c community composi�on & ↑ threat to species’ persistence
Death of some biota
Droughts(see Fig. 3)
↑ Air & water temperature
↑ Risk that upper thermal limits of biota (including
aqua�c, riparian & terrestrial taxa and life stages) are
exceeded
↑ Risk of sub -lethal effects of
hot extremes on biota
Δ Reproduc�on & recruitment
Fig. 2 Effects of heat waves and hot days on rivers, as based on
links among drivers and responses identified in the Australian lit-
erature (see text for detail). Responses may be affected by the inten-
sity, duration and/or timing of heat waves and hot days. Solid
arrows show direct links, broken arrows where one event often
coincides with or increases the likelihood or intensity of the linked
event. Ovals show where effects are hypothesised (event-specific
evidence scarce in the Australian literature). D, change in.
© 2014 John Wiley & Sons Ltd, Freshwater Biology, doi: 10.1111/fwb.12515
4 C. Leigh et al.
experience future droughts of increased duration and
intensity (Risbey, 2011; Steffen et al., 2013). Similar
changes may also occur in parts of Europe, the Mediter-
ranean, the Americas and Africa (IPCC, 2012, 2014).
Ecological effects
Supra-seasonal river droughts are ‘ramp’ disturbances,
increasing in strength and spatial extent over time (Lake,
2003). In the initial stages of drought, riparian habitats
disconnect from channels and littoral habitat dries (Boul-
ton, 2003). Lateral and longitudinal hydrological connec-
tivity is reduced or lost as flow declines, creating
isolated pools (Fig. 3). In downstream river sections and
estuaries, reduced flow can limit the delivery of nutri-
ents from upstream, alter productivity and trophic struc-
ture and increase sediment accumulation (Lake, 2011).
Once surface flows cease, many rivers enter a lentic
phase associated with major changes in water physico-
chemistry (e.g. Sheldon & Fellows, 2010). Pools recede,
and surface water can disappear completely (Fig. 1g).
Declining groundwater levels and increasing salinity in
groundwater-dependent systems and floodplains can
lead to loss of riparian and floodplain vegetation (e.g.
Murray et al., 2003; Fig. 3). In floodplain forests of the
Murray–Darling Basin (MDB), stands of drought-tolerant
Eucalyptus camaldulensis suffered increased mortality
during the Millennium Drought (Horner et al., 2009).
Similarly, drought-related low flows have increased
salinity levels in the lower lakes of the MDB (Mosley
et al., 2012) and contributed to the loss of vulnerable fish
populations (Nannoperca obscura; Wedderburn, Hammer
& Bice, 2012). Ultimately, there is a shift from aquatic to
terrestrial habitat as organisms from surrounding ripar-
ian areas move into dry channels (Steward et al., 2012).
Biotic responses to drought vary depending on many
factors, but in general, organisms that inhabit non-peren-
nial systems have adaptive traits of resistance or resil-
ience to dry periods and may be more likely to survive
supra-seasonal drought than those from perennial sys-
tems (Lake, 2003; Rolls, Leigh & Sheldon, 2012; Fig. 3).
Fish species that tolerated the Millennium Drought in
the MDB tended to have traits associated with adapta-
tion to warm environments (e.g. high spawning temper-
atures), the ability to switch diets and life-history
strategies including delayed maturation, short spawning
seasons and high fecundity (Chessman, 2013). The Mil-
lennium Drought also altered macroinvertebrate com-
munities in Victorian streams, with conditions favouring
taxa tolerant of low flow and poor water quality (Rose,
Metzeling & Catzikiris, 2008; Thomson et al., 2012). Des-
iccation-resistant life-history stages also enable some
taxa (e.g. zooplankton and aquatic plant species, Brock
et al., 2003) to survive drought, although survival can
depend on drought duration. Cladoceran egg banks
from Australian dry land rivers became depleted when
drought lasted more than 6 years, which likely slowed
recovery of microinvertebrate communities and altered
post-drought food webs (Jenkins & Boulton, 2007).
Drought survival depends on the type, quality and
quantity of refugia and the hydrological history and con-
nectivity of habitats (Robson, Chester & Austin, 2011;
Bogan, Boersma & Lytle, 2014). The temporal availability
of refugia and their position in river networks influence
macroinvertebrate community resistance and resilience
to drought (Marshall et al., 2006; Sheldon et al., 2010;
Chester & Robson, 2011; Fig. 3). The rate of water
decline may also affect the ability of some organisms to
find refugia and influence the availability of basal
resources for riverine food webs (Bunn et al., 2006).
↑ Buildup of organic & non-organic material in
dry channels & disconnected pools Heavy rainfall
& floods(post-drought)
(see Fig. 5)
Droughts
↓ Connec�vity of aqua�c habitats
↓Flow & flow permanence
↓ Size &/or availability of
aqua�c habitats & refugia
Groundwater decline
↑ Risk of hypoxic blackwater
events
↑ Risk of fish kills & threat
to other aqua�c taxa
↓ or Δ water quality
↑ Mortality of riparian & floodplain vegeta�on
Δ Community composi�ons (↓sensi�ve & flow-
dependent, ↑ tolerant biota & terrestrial biota)
↓ Influx of nutrients from
upstream to downstream
reaches
Fig. 3 Effects of droughts on rivers, as based on links among driv-
ers and responses identified in the Australian literature (see text for
detail). Responses may be affected by the duration, extent and/or
intensity of droughts. For example, build-up of material on dry
channels and in pools and the risk of hypoxic blackwater events
increase with duration and extent; disconnection among habitats
increases and the size and availability of habitats decline with
increased duration, intensity and extent; survival of aquatic taxa
declines with increased duration. See Fig. 2, for explanation of
arrows, shapes and symbols.
© 2014 John Wiley & Sons Ltd, Freshwater Biology, doi: 10.1111/fwb.12515
Extreme events and riverine ecosystems 5
Knowledge gaps
Although the long-term (decadal) consequences of
supra-seasonal drought are still poorly understood
(Bond, Lake & Arthington, 2008), recent research sug-
gests prolonged events can elicit novel community tra-
jectories and extirpate sensitive species (Bogan & Lytle,
2011; Bogan et al., 2014). However, research is frequently
opportunistic, sporadic and short term (e.g. <1–2 years),
and there is a pressing need for comprehensive monitor-
ing programmes to assess drought impacts and recovery
processes systematically across regions and taxa (Rose
et al., 2008; Lake, 2011; Thomson et al., 2012). Research
typically focuses on macroinvertebrates, fish and ripar-
ian vegetation, with less emphasis on other groups such
as microbes and microinvertebrates. While drought
impacts on the structure of some key assemblages are
well described, consequences for ecosystem processes
and higher levels of biological organisation are less well
known (e.g. Ledger et al., 2013; Woodward et al., 2012).
Incorporating species resistance and resilience traits
into biomonitoring approaches will enhance mechanistic
and predictive understanding of drought impacts (e.g.
Sheldon, Marsh & Rolls, 2012; Chessman, 2013). In many
cases, however, trait information is incomplete or held
in separate repositories and literatures (Crook et al.,
2010). We also need to know how traits interact and bet-
ter understand relationships between traits and biotic
interactions (Verberk, Van Noordwijk & Hildrew, 2013).
For example, a trait such as air breathing that aids sur-
vival of anoxic conditions during drought may increase
predation risk where movement to the water surface
increases encounter rates (Robson et al., 2011). Water-
quality guidelines specific to drought periods are also
required to manage and protect surface and subsurface
drought refugia, thereby supporting species persistence
(e.g. Sheldon & Fellows, 2010).
Fires
Definitions, observations and projections
Much of Australia is vulnerable to wildfire due to the
dry, hot weather and volatile compounds in many of the
native plant species. In the north, the fires generally
occur in winter and spring (the dry season in northern
Australia), whereas in the south, they generally occur in
summer and autumn (BOM, 2009). Hot, dry and windy
conditions increase fire danger (Clarke, Smith & Pitman,
2011), linking heat waves, drought and strong winds to
fire. Since the 1970s, extreme fire weather has increased,
particularly in the south-east, and projections indicate
this trend will continue, with the annual fire season
starting earlier and lasting longer (Lucas et al., 2007;
CSIRO & BOM, 2014). Projections are similar for New
Zealand and increased frequency of fire risk is projected
for much of the Americas (IPCC, 2012).
Ecological effects
One of the most significant and commonly reported
effects of wildfire is the large-scale destruction of
terrestrial vegetation (Fig. 1f). In severely burned and
devegetated catchments, exposed soils become hydro-
phobic, increasing run-off and associated inputs of fine
sediment and nutrients (e.g. Lane, Sheridan & Noske,
2006; Fig. 4). For instance, severe fires in Victoria sub-
stantially elevated stream phosphorus concentrations
and conductivity (Chessman, 1986). Similarly, severe
burning in experimental forests in New South Wales
increased stream nitrate concentrations by an order of
Fires
Destruc�on of catchment &
riparian vegeta�on
Erosion
Fine sediment input
Nutrient input
Burned vegeta�on input
Habitat smothered
Heavy rainfall & floods
(post-fire)(see Fig. 5)
Water quality
Abundance & mortality of
sensi�ve species
Periphytoncover
Composi�on of func�onal feeding
groups of consumers
Aqua�c community composi�on ( sensi�ve,
tolerant species)
Droughts(see Fig. 3)
Heat waves & hot days
(see Fig. 2)
Habitat for riparian terrestrial
fauna & bio�c community
composi�on
Shading
Organic ma�er quality &/or
input
↑
↑ ↑
↑
↑
↓
↓
↓
↓↓Δ Δ
Δ
Δ
↓
Fig. 4 Effects of fires on rivers, as based on links among drivers
and responses identified in the Australian literature (see text for
detail). Responses may be affected by the intensity and/or extent
of fires, for example by increasing the extent and quantity of vege-
tation destroyed or reducing the survival or abundance of species.
See Fig. 2, for explanation of arrows, shapes and symbols.
© 2014 John Wiley & Sons Ltd, Freshwater Biology, doi: 10.1111/fwb.12515
6 C. Leigh et al.
magnitude (Mackay & Robinson, 1987). Changes to
riparian zones may also affect incident light and alter
temperature regimes.
The ecosystem consequences of fire can be exacer-
bated by heavy rains (Cooper et al., 2014), with the com-
bined, interactive impacts of these two extremes creating
a compound event (sensu IPCC, 2012) characterised by
intense sedimentation and turbidity (Fig. 4). In the Cot-
ter River catchment of the Australian Capital Territory, a
large storm 1 month after severe wildfire raised stream
turbidity to an unprecedented level of 3000 NTU (White
et al., 2006). By contrast, fires not followed by heavy rain
tend to have less impact on rivers, as was the case in
Australia’s wet–dry tropics, where fires occurring early
in the dry season had little effect on water quality
(Townsend & Douglas, 2000).
Declines in abundances and richness of instream
fauna are often observed following wildfire (Fig. 4). For
instance, macroinvertebrate communities in the most
severely burned areas of the Cotter River catchment
were impoverished compared with those in unburned
areas (T. Nelson, unpubl. data). However, the scale of
the effects depends on the intensity of fire and post-fire
rainfall (Papas, 1998). A range of conditions in post-fire
streams, including heavy sedimentation and poor water
quality (e.g. dissolved oxygen sags, chemicals from
burned material), has also been associated with reduced
survival and abundance of fish (Carey et al., 2003; Lyon
& O’Connor, 2008).
Fires also affect the availability and quality of trophic
resources. In south-eastern Australian streams, sodium
concentrations increased in burned Eucalyptus viminalis
leaves during post-fire decomposition, and changes in
microbial activity ultimately reduced the amount of bio-
available carbon (P. Love, unpubl. data). In turn, fire-
induced changes in the quality and quantity of trophic
resources can alter the composition of functional feeding
groups (Verkaik et al., 2014; Fig. 4). For example, sub-
stantial inputs of fine suspended sediment after fire in
the Cotter River catchment scoured and smothered
periphyton, reducing the occurrence of herbivorous
scrapers (Peat, Chester & Norris, 2005; T. Nelson, un-
publ. data).
Knowledge gaps
The effects of fire can be immediate and also extend to
the short term (<1 year), mid term (1–10 years) or long
term (>10 years) (Minshall, Brock & Varley, 1989). Least
is known about the long-term ecological consequences of
fire, although there is also some uncertainty regarding
short-term impacts, which may be underestimated when
post-fire sampling is delayed (Verkaik et al., 2014).
Research on the influence of land use on post-fire recov-
ery is rare, even though this likely determines recovery
rates. In the Lower Cotter River catchment, for example,
stream turbidity recovered more quickly in naturally
regenerating, native forest than in plantation pine forest
(Harrison et al., 2014). Research on interactions between
fire and other extreme events such as drought and flood
is also needed. The trajectory of community recovery
from fire during drought periods may be altered by
burning of dewatered benthic sediments (Cowell, Mat-
thews & Lind, 2006) or ash physicochemistry (Verkaik
et al., 2014), but our understanding of such interactions
is incomplete.
Heavy rainfall and floods
Definitions, observations and projections
Heavy rainfall events are periods of prolonged and/or
intense rainfall within a specific location relative to
recorded averages and are often defined by expected
return intervals over decades or centuries (Steffen et al.,
2013). The events are often associated with inland flood-
ing because surface water accumulations and run-off
can rapidly inundate dry lands and floodplains. Austra-
lian indices used to describe these events include heavy
and very heavy precipitation days (≥10 and 30 mm,
respectively) and very wet and extremely wet days
(annual total precipitation when daily precipitation
>95th and 99th percentiles, respectively) (BOM, 2014b).
Several of these extremes occurred across eastern Aus-
tralia in 2010–2012, breaking the Millennium Drought.
Severe flooding occurred in central and southern
Queensland in 2010–2011, during a period including the
state’s wettest December on record (NCC BOM, 2011;
Fig. 1e). Both central and northern Australia experienced
the wettest dry season on record in 2010 and record
heavy rainfall and flooding occurred in most states and
territories in 2010, 2011 and 2012 (NCC BOM, 2011,
2012; BOM, 2012a). Heavy rainfall intensity is expected
to increase in Australia in the 21st century, whereas the
number of events may increase, decrease or remain
unchanged depending on region (Alexander & Arblast-
er, 2009), matching projections elsewhere (IPCC, 2012;
Donat et al., 2013). Such changes in rainfall patterns will
likely affect flood regimes, altering the intensity, dura-
tion, frequency and timing of events.
© 2014 John Wiley & Sons Ltd, Freshwater Biology, doi: 10.1111/fwb.12515
Extreme events and riverine ecosystems 7
Ecological effects
The ecological importance of floods in riverine ecosys-
tems is well documented, both within Australia and
internationally. Floods influence ecosystem processes,
affecting within-channel productivity, particulate organic
matter retention, decomposition, nutrient transforma-
tions and microbial processes (Baldwin & Mitchell, 2000;
Hladyz et al., 2012). Floods also affect terrestrial flood-
plain biota (Horrocks et al., 2012) and influence surface
water and groundwater quality and recharge (Cend�on
et al., 2010) (Fig. 5). Rising waters increase hydrological
connectivity at various spatial scales, particularly in arid,
semi-arid and wet–dry zones (Douglas, Bunn & Davies,
2005; Leigh et al., 2010). This facilitates transport of
materials, such as propagules, nutrients, organic matter
and woody debris, both downstream and to adjacent
floodplains and riparian zones (Ballinger & Lake, 2006;
Erskine et al., 2012; Fig. 5). Floods may also cause
siltation and ‘sand slugs’, alter channel morphology,
scour substrata, remove in-situ basal resources such as
macrophytes and detritus and translocate, injure or kill
biota (Lake, 2000; Fig. 5).
Much research has focused on flood-driven hydrologi-
cal connectivity and food webs. Floods drive spatial sub-
sidies in food resources and affect the relative
importance of autochthonous and allochthonous carbon
sources (Bunn et al., 2006; Reid et al., 2008). In some
cases, mobile consumers such as fish and birds may
transfer resources between habitats connected by inun-
dation (Jardine et al., 2012). River–floodplain connections
can also facilitate ‘boom-bust’ cycles in basal productiv-
ity and invertebrate emergence, with flow-on effects to
other components of the food web (Boulton & Lloyd,
1992; Bunn et al., 2006; Fig. 5). The floods of 2010–2011
increased taxonomic richness and abundance of zoo-
plankton in several southern-MDB rivers (Ning et al.,
2013). In central Australia, post-flood booms in fish pro-
duction subsequently fuelled high levels of heterotrophic
bacterial production once waters (and fish) retracted to
isolated waterholes (Burford et al., 2008).
Floods also alter water quality. Hypoxic blackwater
events can result from the microbial breakdown of
deposited or leached organic matter, although their
intensity and extent are often exacerbated by pre-flood
drought and human activities (Whitworth, Baldwin &
Kerr, 2012; Figs 4 & 5). As the Millenium Drought was
breaking in late 2010, one such event killed many native
crustaceans and fish and led to the emergence of native
crayfish from affected waters (King, Tonkin & Lieshcke,
2012; McCarthy et al., 2014). However, non-native fish
such as Cyprinus carpio were apparently unaffected.
Floods can facilitate the spread of non-native and
invasive species. Widespread flooding has probably
facilitated the spread of Phyla canescens, an invasive
riparian plant, across eastern Australia (Murray, Stokes
& Van Klinken, 2011; Fig. 5). By contrast, remnant popu-
lations of native freshwater mussels are threatened by
extreme floods, particularly where events exacerbate
existing impacts of catchment degradation (Jones & By-
rne, 2010). However, accumulation of large woody deb-
ris during flooding can facilitate post-flood recovery of
native riparian vegetation (e.g. in Australia’s semi-arid
savannah; Pettit & Naiman, 2005) and, in some cases,
floods favour native over non-native species (Costelloe
et al., 2010; Ho, Bond & Thompson, 2013). In south-east-
ern Australia, flooding increased species richness of
native plants in E. camaldulensis stands, with twice as
Heavy rainfall & floods
Catchment & channel erosion
Movement of large amounts of
organic & inorganic ma�er (including biota)
Booms in produc�on & trophic interac�ons
Spread of (e.g. invasive)
species
Groundwater recharge
Water quality
Watering of riparian & floodplain vegeta�on
Connec�vity of aqua�c habitats
Triggers dispersal,
spawning & recruitment
opportuni�esDroughts
(pre-flood)(see Fig. 3)
Tropical cyclones
(see Fig. 6)
Risk of hypoxic blackwater
events
& threat to other aqua�c taxa
Community composi�on ( sensi�ve, tolerant biota)
↑ ↑
↑↑
↑ Risk of fish kills
↑↓Δ
Δ
Δ
Fig. 5 Effects of heavy rainfall and floods on rivers, as based on
links among drivers and responses identified in the Australian lit-
erature (see text for detail). Responses may be affected by the inten-
sity, timing and/or extent of these events. For example, the extent
of habitat connectivity and amount of channel erosion and scouring
increase as these events increase in intensity and extent; timing of
these events relative to drought will affect the risk of hypoxic
blackwater events; timing and intensity of events relative to life-his-
tory stages and age of biota may affect the chance of translocation,
injury or death. See Fig. 2, for explanation of arrows, shapes and
symbols.
© 2014 John Wiley & Sons Ltd, Freshwater Biology, doi: 10.1111/fwb.12515
8 C. Leigh et al.
many natives found in flooded as unflooded areas
(Horner et al., 2012).
Knowledge gaps
Floods that are aseasonal, recurrent or interact with
other extreme events (e.g. fire and drought) or land-use
degradation, can markedly affect river biodiversity.
However, explicit studies of such phenomena are scarce.
The timing of heavy rainfall and extreme floods relative
to key stages of aquatic life cycles may influence recruit-
ment, dispersal and foraging opportunities (e.g. in fish,
Humphries, King & Koehn, 1999), and shifts in flood fre-
quency and intensity may affect the persistence or
spread of invasive species, for example by providing
opportunities for passive or active dispersal within and
between river networks. However, more research is
needed to determine the vulnerability and dispersibility
of invasive species during floods. Impacts of extreme
floods on genetic diversity of threatened populations are
also uncertain, although it has been proposed that more
large-scale floods could increase gene flow, while more
intense floods could create population bottlenecks (Wor-
thington Wilmer et al., 2011). Research on ecological out-
comes of environmental flows featuring managed
flooding may help us understand impacts of future flood
regimes, but existing data are biased towards particular
taxa (e.g. fish and floodplain vegetation) and systems
(e.g. MDB rivers; Aldous et al., 2011). We require more
rigorous, high-resolution hydrological models (Pittock
et al., 2006), and these must be better integrated with
ecological models to better predict relationships between
changing rainfall, future flood regimes and ecological
responses.
Tropical cyclones
Definitions, observations and projections
Tropical cyclones are low-pressure systems that develop
over warm, tropical marine-waters and are associated
with gale-force winds (sustained at ≥63 km h�1 or in
gusts >90 km h�1) (BOM, 2014c). Winds sustained at
≥118 km h�1 (or gusts >165 km h�1) qualify events as
severe tropical cyclones (STCs), also known as hurri-
canes or typhoons. Well-documented STCs of the last
50 years in Australia include Tracy, Larry (Fig. 1d),
Monica and Yasi (BOM, 2014d). STC Monica was associ-
ated with maximum recorded winds of 130 km h�1 and
rainfall of up to 340 mm in 24 h. Although no clear
trends in tropical cyclone characteristics have been
found in the Australian region, projections for tropical
zones in Australia and beyond suggest they may become
less frequent but more intense (CSIRO & BOM, 2012;
IPCC, 2012). The proportion of severe events is therefore
likely to increase.
Ecological effects
As with fire, one of the most commonly reported effects
of tropical cyclones is vegetation damage, with riparian
zone vegetation being particularly susceptible (Fig. 6).
In the forests of Magela and Ngarradj Creek catch-
ments, Northern Territory, wind gusts during STC
Monica created significant treefall and riparian zones
sustained greater damage than did other areas (Saynor
& Erskine, 2008; Staben & Evans, 2008). In some parts
of the Ngarradj Creek catchment, the entire riparian
rainforest was destroyed. STC Monica occurred at the
end of the tropical wet season when riparian zone soils
were saturated and streams inundated, so these factors
probably exacerbated the damage (Saynor & Erskine,
2008; Fig. 6).
Riparian vegetation was also damaged during STC
Larry (Fig. 1d). Six months after that event, pioneer spe-
cies in north-east Queensland rainforests were more
severely damaged than other species. They suffered dis-
proportionately more uprooting, particularly along
stream edges (Pohlman, Goosem & Turton, 2008). This
was associated with sandy soils near streams, producing
loose root systems, and bank erosion during post-cyclo-
nic floods. However, while damage to riparian vegeta-
tion can be substantial, the associated delivery of large
woody debris to rivers contributes important habitat
and increases channel complexity (Erskine et al., 2012;
Fig. 6). After STC Monica in the Ngarradj Creek catch-
ment, the number of pieces of large wood per metre of
channel more than doubled (Erskine et al., 2012).
Tropical cyclones producing heavy rainfall change
water quality in rivers (Fig. 6). Rainfall following Tropi-
cal Cyclone Celeste in north-east Queensland (January
1996) rapidly increased water level and discharge in the
Burdekin River (Alexander et al., 2001). River water
became highly turbid, with suspended solids concentra-
tions >4000 mg L�1. Conductivity decreased rapidly but
recovered to pre-event conditions within 1–2 weeks. In
contrast, increased suspended solids concentrations were
observed up to a year following STC Monica (Evans &
Moliere, 2010). Differences in the effects of Celeste and
Monica on water quality were likely influenced by dif-
ferences in their timing relative to the first, flushing
flows of the wet season (Alexander et al., 2001), their
© 2014 John Wiley & Sons Ltd, Freshwater Biology, doi: 10.1111/fwb.12515
Extreme events and riverine ecosystems 9
intensity (e.g. erosive forces on soils and sediments) and
the amount of damage caused to riparian vegetation
(Fig. 6).
Recharge of water stores in northern Australia and
aquifers in central Australia relies heavily on tropical
cyclones, and thus recharge can be sporadic depending
on event timing, frequency and intensity (Barron et al.,
2011; McGrath et al., 2012; Fig. 6). Past long-term
declines in tropical cyclone frequency are associated
with ‘water storage droughts’ and desertification (Nott,
2011; McGrath et al., 2012). Tropical cyclones in northern
Australia also deliver rains to more southerly regions as
they degenerate to extropical lows during their overland
trajectories. These rains supply water that produces
flows and sustains biota in temporary wetlands of dry-
land and semi-arid river systems (Roshier et al., 2001)
and in catchments that discharge into the Great Barrier
Reef lagoon (Boer, 2010).
Knowledge gaps
Much of our knowledge of tropical cyclone effects is
drawn from vegetation studies. We know little about
how they affect instream biota (e.g. fish, invertebrates;
although see Mallin et al., 1999) and ecosystem pro-
cesses, either directly or via indirect effects on riparian
zones and water quality (Fig. 6). Although we have
some evidence suggesting cyclone (and flood) timing
affects ecological responses (e.g. Alexander et al., 2001;
Saynor & Erskine, 2008), our understanding of this
aspect and cyclone–flood interactions is quite limited.
Moreover, the lack of studies combining pre- and
post-event data limits understanding of impacts and
recovery.
Storm surges and coastal flooding
Definitions, observations and projections
Coastal flooding results from high water levels pro-
duced by sea-level fluctuations during short-term,
localised storm-surge and wave events, and longer term
sea-level rise (Eliot, 2012). The primary cause of surge-
related coastal flooding is the landfall of low-pressure
systems with strong winds (CSIRO & BOM, 2007).
More intense events occur when storm surge coincides
with high tides, and events become more frequent as
sea level rises (S�anchez-Arcilla et al., 2008; Fig. 7).
Surge-related coastal flooding can occur anywhere
topography allows (CSIRO & BOM, 2007; BOM, 2012b).
However, projected changes in storm-surge and inun-
dation impacts vary regionally and can depend on the
method by which storm-tide intervals are estimated
(CSIRO & BOM, 2007). It is very likely that mean sea-
level rise will continue to contribute to global upward
trends in extreme coastal high water levels (IPCC,
2012).
Heavy rainfall & floods(see Fig. 5)
Damage to catchment &
especially riparian vegeta�on
Groundwater recharge
Intensity of winds &/orwind gusts
Erosion
Fine sediment, organic ma�er &
nutrient input
Water quality
Tropical cyclones
Delivery of large woody
debris to streams
Storm surges & coastal flooding
(see Fig. 7)
Sandy riparian
soils
Satura�on of riparian
soils
Intensity & extent of stream
& floodplain inunda�on
Aqua�c community composi�on
Instream habitat for aqua�c biota &
channel complexity
Diversity of fish & macroinvertebrates
Shading
Wetland replenishment
↑
↑
↑
↑
↑
↑
↑
↑
↓
Δ
Δ
↑
↑
Fig. 6 Effects of tropical cyclones on rivers, as based on links
among drivers and responses identified in the Australian literature
(see text for detail). Responses may be affected by the intensity,
timing and/or frequency of these events. For example, saturated
soils are more likely during wet-season events; events before the
wet season’s first flushing flows increase inputs of sediment,
organic matter and nutrients to streams and the time taken to
return to pre-event conditions; the timing and frequency of events
affect the sporadicity of groundwater recharge. See Fig. 2, for
explanation of arrows, shapes and symbols.
© 2014 John Wiley & Sons Ltd, Freshwater Biology, doi: 10.1111/fwb.12515
10 C. Leigh et al.
Ecological effects
Storm surges alter habitats by pushing sea water into
estuaries, brackish lagoons and wetlands, which in turn
can redistribute sediment, contribute to erosion (Cahoon,
2006; Howes et al., 2010) and force saline water into fresh-
water river sections (CSIRO & Bureau of Meteorology
(BOM), 2007; Kang & King, 2012; Fig. 7). Surge-related
effects of increased salinity and geomorphological
changes on riverine biota (e.g. aquatic macroinverte-
brates) are, however, understudied (Kang & King, 2012),
and most research has been performed outside Australia,
primarily on aquatic macrophytes (e.g. Frazer et al., 2006).
Knowledge gaps
Effects of storm surge and coastal flooding on riverine
ecosystems are poorly known in Australia and globally.
Studies from North America suggest aquatic vegetation
in coastal fresh waters is susceptible to salinity changes
and erosion from storm surge (e.g. Frazer et al., 2006).
However, these studies have focussed on wetlands, and
their applicability to rivers is unclear (Fig. 7). Responses
to coastal flooding may also differ among and within
taxa, but again, knowledge is incomplete. While short-
term shifts in fish community composition were
observed following saltwater surge into freshwater
reaches of rivers near Hurricane Katrina’s landfall zone
(Schaefer et al., 2006), amphibian species in freshwater
coastal wetlands showed resistance to water chemistry
changes (Gunzburger et al., 2010). Recovery of freshwa-
ter fauna following saltwater flooding is likely influ-
enced by dispersal characteristics, with active dispersal
(e.g. swimming) contributing to rapid recovery (Watana-
be et al., 2014).
Understanding of storm-surge effects in rivers could
be informed by knowledge of species salinity tolerances,
and by studies of tsunamis (e.g. Watanabe et al., 2014) or
salinisation from progressive saltwater intrusion and
sea-level rise. However, ramp disturbances such as pro-
gressive salinisation may have different impacts from
pulsed events like storm surges, where recovery to pre-
disturbance (freshwater) conditions can occur rapidly.
On the other hand, storm surges can rework sediments
and channels and may even cause mass mortalities
requiring long recovery periods (Cahoon, 2006). Intru-
sion of saline waters into ground water during surges
may also prolong recovery of aquatic biota in groundwa-
ter-fed rivers (Fig. 7). Furthermore, surge-induced flood-
ing may be followed by highly turbid floods (of inland
origin) associated with the same cyclone or storm (e.g.
Gong & Shen, 2009), causing abrupt changes from fresh-
water to saline water to turbid freshwater conditions.
Future directions and concluding remarks
Extreme events are locally and globally important, with
impacts often disproportionate to their duration (Smith,
2011). The ecological effects of extremes can be difficult
to determine from opportunistic studies, and more
research is needed to develop a sound mechanistic
understanding of responses to inform prediction of
future events, where they occur singly or in combina-
tion. Among event types, research on floods, droughts
and fires is more extensive than for heat waves, tropical
cyclones and storm surges. While research on all events
is needed, these latter events remain a particular priority
for future research.
As the frequency of extreme events increases, the his-
tory of past events will become increasingly important in
shaping responses to subsequent extremes (IPCC, 2012).
Compound events will be particularly deleterious, as has
been observed where heavy rains and flooding follow fire
or drought. The intensification and coincidence of
extreme events may elicit regime shifts, novel interactions
and/or abrupt transitions in species distributions and
Heavy rainfall & floods
(post-surge)(see Fig. 5)
Storm surges & coastal flooding
Death or of saline-intolerant species, &/or
composi�on
Sediment redistribu�on &
Abrupt salinity in lowland river
sec�ons (fresh to saline to fresh)
Intrusion of saline water into groundwater &
groundwater-dependent
riversWater
quality
Tropical cyclones (see Fig. 6)
Sea-level rise
High �des
↑ salinity in ↑ erosion
Δ
Δ aqua�c community
Δ
↓ abundance
Fig. 7 Effects of storm surges and coastal flooding on rivers, as
based on links among drivers and responses identified in the Aus-
tralian literature (see text for detail). See Fig. 2, for explanation of
arrows, shapes and symbols.
© 2014 John Wiley & Sons Ltd, Freshwater Biology, doi: 10.1111/fwb.12515
Extreme events and riverine ecosystems 11
Table
1Hypotheses
andpotential
approaches
toexplore
theim
pactofextrem
eclim
atic
even
tsonfreshwater
hab
itat
andacross
levelsofbiological
organ
isation
Even
tsan
dfuture
projections
Hypotheses*
Approaches*
Heatwav
esan
dhot
day
swillincrease
innumber
andduration
Habitat
Increasedim
portan
ceofcool-water
refuges
forheat-sensitivetaxa
Species
andpopu
lations
More
freq
uen
texceed
ance
ofupper
thermal
tolerance
limitsofbiota
Red
ucedresilien
ce(recovery)ofheat-sensitivepopulations
Localextinctionorcontractionin
distributionsofheat-sensitivetaxa
Com
munities
Loss
offavourable
hab
itat
andsp
eciesan
dim
migrationofnew
specieswillaltercommunity
structure
Functioning
Organ
icmatterprocessingmay
alterdueto
localextinctionsan
dbeh
aviouraleffectsofheatstress
on
primaryconsu
mers
Measu
remen
tofhab
itat
characteristicsto
iden
tify
thermal
refugia
Experim
ents
applyinghightemperature
extrem
esofdifferentdurationsan
dfreq
uen
cies,
downscalingclim
atemodelsto
gen
eraterealistic
temperature
extrem
esan
dregim
esModellingto
predictch
anges
indistributionsof
heat-sensitivetaxa,
particu
larlyforknown
vulnerab
letaxa
Droughts
willincrease
inintensity,extent
andduration
Habitat
andevents
Increasedextentofhydrological
disconnection(terrestrialisation);hab
itat
loss,includingdrought
refugia;increasedim
portan
ceofremainingrefugia
Increasedinciden
ceofdroughts
interactingwithother
extrem
eev
ents
(compoundev
ents)
Species
andpopu
lations
Loss
orcontractionin
distributionsofvulnerab
letaxa(includingflow-dep
endan
ttaxa)
Com
munities
Chan
ges
inrich
nessan
dcommunitycompositionresu
ltingfrom
chan
ges
inhab
itat
and/orlocal
speciesextinctions,
includingloss
offlow-dep
enden
ttaxafrom
stream
networksan
dch
anges
inveg
etationtype,
andincreasedlikelihoodofnovel
communitytrajectories
Functioning
Accumulationoforgan
icmatterondry
stream
bed
s;increasedoccurren
ceofhypoxic
blackwater
even
ts;alteredorgan
icmatterprocessingratesin
stream
s;increasedfuel
load
foran
drisk
offire;
potentially
drivingsh
ifts
inbeh
aviour(e.g.foraging)an
dcompositionofterrestrialbiota
Extended
periodsofreducednutrientinputinto
downstream
(e.g.lowland)reaches
Chan
ges
infood-w
ebstructure
anddynam
icsvia
loss
ofrare
taxaan
dpredators
Experim
ents
applyingdifferenthydrological
(dis)
connectivitytreatm
ents
anddryingdisturban
ces
ofdifferentintensities
over
multiple
spatiotemporalscales
Experim
ents
applying‘droughtdisturban
ces’
incombinationwithother
extrem
eev
enttreatm
ents
Experim
ents
ondry-stream
organ
icmatter
break
down,pre-conditioningan
dconsu
mptionor
use
bybiota
Modellingto
predictch
anges
inav
ailabilityan
dconfigurationofdroughtrefugia
andtheir
accessibilityto
vulnerab
letaxa
Fires
willincrease
infreq
uen
cy,therisk
of
fire
willincrease,an
dan
nual
fire
seasons
willstartearlieran
dlast
longer
Habitat
andevents
Accumulationoffinesedim
entonstream
bed
san
dreductionin
stream
hab
itat
size
andquality
Increasedexposu
reofstream
chan
nelsto
sunfrom
decreasein
canopycover,increasingstream
temperaturesan
dexacerbatingeffectsofheatwav
esan
dhotday
sIncreasedinciden
ceoffire–fl
oodinteractions(compoundev
ents)
Species
andpopu
lations
Loss
orcontractionin
distributionsofheat-sensitivetaxaan
dthose
sensitiveto
finesedim
ent
accu
mulation
Com
munities
Chan
ges
inrich
nessan
dcommunitycompositionresu
ltingfrom
chan
ges
inhab
itat,localsp
ecies
extinctionsan
d/orresp
onsesto
chan
ges
inorgan
icmatterqualityan
dquality(see
Functioning)
Chan
ges
inveg
etationtypean
dstructure
inriparianzo
nes
Functioning
Increasedquan
tity
ofrefractory
carbon(charcoal)an
dreducedqualityoforgan
icmatterin
stream
san
dresu
ltan
tch
anges
tofoodweb
s
Experim
ents
man
ipulatingthequan
tity
andquality
oforgan
icmatterin
stream
sExperim
ents
man
ipulatingsedim
entan
dash
inputs
under
both
dry-an
dwet-stream
conditions
Modellingto
predictextentofsedim
ent
accu
mulationan
dtran
sport
instream
s,including
regionswithvulnerab
letaxa,
communitiesan
d/
orecosystem
s
© 2014 John Wiley & Sons Ltd, Freshwater Biology, doi: 10.1111/fwb.12515
12 C. Leigh et al.
Tab
le1
(Continued
)
Even
tsan
dfuture
projections
Hypotheses*
Approaches*
Heavyrainfall(and
floods)
willincrease
innumber
and
intensity
Habitat
andevents
Increasedfreq
uen
cy,intensity
andinundationextentan
dnovel
timingoffloods(hydrological
connectionev
ents)
Increasedoccurren
ceofsu
ccessivefloodsan
dfloodsoccurringin
combinationwithother
extrem
eev
ents
(compoundev
ents)
Increasedoccurren
cesofch
annel
erosion
Species
andpopu
lations
Increasedfreq
uen
cyof‘hab
itat-connecting’ev
ents
willfacilitate
spread
ofsp
ecies,
including
invasives,an
dthetran
sport
ofother
materials
(organ
ican
dinorgan
ic)
Novel
timingofev
ents
may
trigger
spaw
ningcu
esin
somesp
eciespossibly
lead
ingto
unsu
ccessful
recruitmen
t,forexam
ple
ifsp
awningoccurs
when
seasonal
conditions(e.g.temperatures)
arenot
suitab
lefordev
elopmen
tCom
munities
Chan
ges
inrich
nessan
dcommunitycompositionresu
ltingfrom
chan
ges
inhab
itat,localsp
ecies
extinctionsan
d/orinvasions
Functioning
Increasedtran
sport
oforgan
ican
dinorgan
icmaterials
within
andbetweencatchmen
ts
Experim
ents
applying‘flooddisturban
ces’
successivelyan
d/orin
combinationwithother
extrem
eev
enttreatm
ents
Experim
ents
applyingdifferenthydrological
connectivitytreatm
ents
over
multiple
spatial
scales
Modellingto
predictch
anges
inrecruitmen
tsu
ccessan
ddistributionsofvulnerab
letaxaan
dinvasives
Tropical
cyclones
will
increase
inintensity
Habitat
Increaseddestructionofriparianveg
etation(treefallan
dother
winddam
age)
andincreaseddelivery
oflargewoodydeb
risto
stream
sIncreasedsh
elteringhab
itat
forfish
andmacroinvertebratesan
dincreasedsu
bstrate
forbiofilm
growth
(foodforconsu
mers)
Increasedintensity
ofwater-qualitych
anges
followingev
ents
(e.g.extrem
eincreasesin
turbidity),
withprolonged
recovery
Species
andpopu
lations
Creationof(tem
porarily)unsu
itab
lehab
itat
forturbidity-sen
sitiveaq
uatic
biota,potentially
causing
localextinctions
Substan
tial
dam
ageto
orloss
ofwind-sen
sitivetaxain
riparianzo
nes
andincreasedrisk
ofdam
age
towind-toleranttaxa
Com
munities
Chan
ges
inrich
nessan
dcommunitycompositionresu
ltingfrom
chan
ges
inhab
itat
and/orlocal
extinctions
Functioning
Altered
food-w
ebdynam
icsresu
ltingfrom
chan
ges
inriparianveg
etationan
dorgan
icmatterinputs
Experim
ents
applying‘w
indtreatm
ents’of
differentstrength
anddurationunder
high-flow
andcontrol-flow
conditions
Experim
ents
andsu
rvey
sto
establish
how
loss
and
recoveryofriparianveg
etation,an
dch
anges
inturbidity,alterfoodweb
san
daq
uatic
community
composition
Modellingto
predictextentan
ddurationof
turbidityan
dother
water-qualitych
anges
during
andafterev
ents
ofdifferentintensity
Storm
surges
will
increase
infreq
uen
cyan
dcoastalflooding
willincrease
inextent
Habitat
Increasedfreq
uen
cyofab
ruptch
anges
insalinityin
freshwater
sectionsofriversan
dincreased
likelihoodofev
ents
affectingsectionsfurther
upstream
than
previouslyknown
Increasedtemporalan
dsp
atialvariabilityin
chan
nel
morphology
Species
andpopu
lations
Loss
orcontractionin
distributionsofsalinity-sen
sitivetaxa
Com
munities
Terrestrial
andaq
uatic
communitiesofaffected
riversbecomemore
similar
tothose
typically
found
inestuarinehab
itats
Experim
ents
applyingab
ruptwater-quality
chan
ges
Modellingto
predictextentan
dpersisten
ceof
saltwater
inundation,particu
larlyin
regionswith
vulnerab
letaxa,
communitiesan
d/orecosystem
s
*Response
variablesin
relationto
thesehypotheses
andcorrespondingap
proaches
may
includebutarenotrestricted
toaerial
andaq
uatic
stag
esofaq
uatic
macroinvertebrates;
fish
;
terrestrialan
daq
uatic
plants;microbialcommunities;
foodweb
s;leaf-litterbreak
downratesan
dorgan
icmatterprocessingdynam
ics;
water-qualityparam
eters;
availability,quality
andpositionofrefugia.
© 2014 John Wiley & Sons Ltd, Freshwater Biology, doi: 10.1111/fwb.12515
Extreme events and riverine ecosystems 13
ecosystem functioning (IPCC, 2014). Successive events of
the same type may alter the organisation of food webs
and disrupt the successional development of biological
communities (e.g. Ledger et al., 2012, 2013) or initiate
novel development trajectories (Turner et al., 1998).
Understanding of compound events and long-term
effects of extreme events could be enhanced by exploit-
ing long-term data collected by monitoring programs for
other purposes such as bioassessment but which capture
pre- and post-event information (e.g. Rose et al., 2008).
Standardisation and maintenance of monitoring pro-
grams across regions, along with question-driven pro-
grams and adaptive designs would also improve the
utility of long-term datasets to elucidate mechanisms
underlying ecological responses to and recovery from
extreme events (Lindenmayer & Likens, 2010). While
such survey data are important, it is clearly impractical
to rely solely on phenomenological research of rare
unpredictable events. A range of experimental
approaches is also required to test hypotheses and estab-
lish a solid, mechanistic understanding of the effects of
extreme events in fresh waters (cf. Wernberg, Smale &
Thomsen, 2012; Stewart et al., 2013b; Table 1).
Extreme events are key elements of the natural vari-
ability that shapes the ecology of rivers and streams.
Freshwater organisms can recover from disturbances,
including extreme events, yet an intensification of distur-
bance regimes in a warming world could exceed the
capacity of communities to rebound. Because future
events are likely to be more extreme than those today
(unprecedented events) and more frequent or novel
combinations are projected (IPCC, 2012), there is an
increased likelihood that extreme events will push some
species or ecological processes beyond critical thresh-
olds, threatening species persistence, altering ecosystem
functioning or tipping communities and ecosystems into
novel states (Paine et al., 1998; Table 1). As the resilience
of aquatic organisms is lessened by a range of anthropo-
genic stressors, the effects of extreme events on riverine
ecosystems must be considered alongside those of
human activities (e.g. Balcombe et al., 2011; Leigh et al.,
2013; Reich & Lake, 2014). As extreme events also occur
against a backdrop of gradual climate change, outcomes
of future events must be considered in the light of these
longer term trends in climate, especially progressive
warming. Studies that apply experimental extreme
events and monitor long-term recovery patterns, apply
experimental extreme events repeatedly over realistic
timeframes (e.g. Ledger et al., 2011), apply experimental
treatments that incorporate extreme events into future
climate scenarios (e.g. Thompson et al., 2013b) or assess
the combined effects of multiple stressors (e.g. Lunt, Jan-
sen & Binns, 2012) will help fill knowledge gaps and
better enable us to predict responses to the events and
novel regimes of the future.
Acknowledgments
We thank Pettina Love, Tom Nelson and Anthea Flo-
rance for information on fires from their Honours
research at the University of Canberra; Steve Turton,
Campbell Clarke and Tom Nelson for photographs; and
Andrew Boulton, Sarah Boulter, Dave Strayer and two
anonymous reviewers for their comments and edits that
improved the manuscript.
References
Aldous A., Fitzsimons J., Richter B. & Bach L. (2011)
Droughts, floods and freshwater ecosystems: evaluating
climate change impacts and developing adaptation strate-
gies. Marine and Freshwater Research, 62, 223–231.
Alexander J., Fielding C.R., Wakefield S.J., George M.T. &
Cottnam C.F. (2001) Fluvial geochemistry through a
short-duration, tropical-cyclone induced discharge event
in the Burdekin River and Hann Creek, North Queens-
land, Australia. Aquatic Geochemistry, 7, 275–293.
Alexander L.V. & Arblaster J.M. (2009) Assessing trends in
observed and modelled climate extremes over Australia
in relation to future projections. International Journal of
Climatology, 29, 417–435.
Balcombe S.R., Sheldon F., Capon S.J., Bond N.R., Hadwen
W.L., Marsh N. et al. (2011) Climate-change threats to
native fish in degraded rivers and floodplains of the Mur-
ray–Darling Basin, Australia. Marine and Freshwater
Research, 62, 1099–1114.
Baldwin D.S. & Mitchell A.M. (2000) The effects of drying
and re-flooding on the sediment and soil nutrient dynam-
ics of lowland river-floodplain systems: a synthesis. Regu-
lated Rivers: Research and Management, 16, 457–467.
Ballinger A. & Lake P.S. (2006) Energy and nutrient fluxes
from rivers and streams into terrestrial food webs. Marine
and Freshwater Research, 57, 15–28.
Barron O.V., Crosbie R.S., Charles S.P., Dawes W.R., Ali R. &
Evans W.R. et al. (2011) Climate change impact on ground-
water resources in Australia. Waterlines report series no
67. National Water Commission, Canberra, Australia.
Beitinger T.L., Bennett W.A. & McCauley R.W. (2000) Tem-
perature tolerances of North American freshwater fishes
exposed to dynamic changes in temperature. Environmen-
tal Biology of Fishes, 58, 237–275.
Boer H. (2010) Policy options for, and constraints on, effec-
tive adaptation for rivers and wetlands in northeast
Queensland. Australasian Journal of Environmental Manage-
ment, 17, 154–164.
© 2014 John Wiley & Sons Ltd, Freshwater Biology, doi: 10.1111/fwb.12515
14 C. Leigh et al.
Bogan M.T., Boersma K.S. & Lytle D.A. (2014) Resistance
and resilience of invertebrate communities to seasonal
and supraseasonal drought in arid-land headwater
streams Freshwater Biology, doi:10.1111/fwb.12522, in
press.
Bogan M.T. & Lytle D.A. (2011) Severe drought drives
novel community trajectories in desert stream pools.
Freshwater Biology, 56, 2070–2081.
Bogan T., Othmer J., Mohseni O. & Stefan H. (2006) Esti-
mating extreme stream temperatures by the standard
deviate method. Journal of Hydrology, 317, 173–189.
Bond N.R., Lake P.S. & Arthington A.H. (2008) The impact
of drought on freshwater ecosystems: an Australian per-
spective. Hydrobiologia, 600, 3–16.
Boulton A.J. (2003) Parallels and contrasts in the effects of
drought on stream macroinvertebrate assemblages. Fresh-
water Biology, 48, 1173–1185.
Boulton A.J. & Lloyd L.N. (1992) Flooding frequency and
invertebrate emergence from dry floodplain sediments of
the River Murray, Australia. Regulated Rivers: Research and
Management, 7, 137–151.
Brock M.A., Nielsen D.L., Shiel R.J., Green J.D. & Langley
J.D. (2003) Drought and aquatic community resilience:
the role of eggs and seeds in sediments of temporary
wetlands. Freshwater Biology, 48, 1207–1218.
Bunn S.E., Thoms M.C., Hamilton S.K. & Capon S.J. (2006)
Flow variability in dryland rivers: boom, bust and the bits
in between. River Research and Applications, 22, 179–186.
Bureau of Meteorology (BOM) (2009) Bushfire weather.
Accessed online 30 July 2014 at: www.bom.gov.au/
weather-services/bushfire/about-bushfire-weather.shtml
Bureau of Meteorology (BOM) (2012a) Record-breaking La
Ni~na events: an analysis of the La Ni~na life cycle and the
impacts and significance of the 2010–11 and 2011–12 La
Ni~na events in Australia. Bureau of Meteorology, Mel-
bourne, Australia.
Bureau of Meteorology (BOM) (2012b) A significant winter
wind and storm surge event in southwest Western Aus-
tralia in early June 2012. Special climate statement 40.
Bureau of Meteorology, Melbourne, Australia.
Bureau of Meteorology (BOM) (2013) Special Climate State-
ment 43 – extreme heat in January 2013. Bureau of Mete-
orology, Melbourne, Australia.
Bureau of Meteorology (BOM) (2014a) About the pilot heat-
wave forecast. Accessed online 30 July 2014 at: www.bom.
gov.au/weather-services/about/heatwave-forecast.shtml
Bureau of Meteorology (BOM) (2014b) About the climate
extremes analyses. Accessed online 30 July 2014 at: www.
bom.gov.au/climate/change/about/extremes.shtml
Bureau of Meteorology (BOM) (2014c) About tropical
cyclones. Accessed online 30 July 2014 at: www.bom.gov.
au/cyclone/about/
Bureau of Meteorology (BOM) (2014d) Previous tropical
cyclones. Accessed online 30 July 2014 at: www.bom.gov.
au/cyclone/history/
Burford M.A., Cook A.J., Fellows C.S., Balcombe S.R. &
Bunn S.E. (2008) Sources of carbon fuelling production in
an arid floodplain river. Marine and Freshwater Research,
59, 224–234.
Cahoon D.R. (2006) A review of major storm impacts on
coastal wetland elevations. Estuaries and Coasts, 29, 889–
898.
Caissie D. (2006) The thermal regime of rivers: a review.
Freshwater Biology, 51, 1389–1406.
Carey A., Evans M., Hann P., Lintermans M., Macdonald T.
& Onnay P. et al. (2003) Wildfires in the ACT 2003: report
on initial impacts on natural ecosystems. Environment
ACT, Canberra, Australia.
Cend�on D.I., Larsen J.R., Jones B.G., Nanson G.C., Rickl-
eman D., Hankin S.I. et al. (2010) Freshwater recharge
into a shallow saline groundwater system, Cooper Creek
floodplain, Queensland, Australia. Journal of Hydrology,
392, 150–163.
Chessman B.C. (1986) Impact of the 1983 wildfires on river
water quality in East Gippsland, Victoria. Australian Jour-
nal of Marine and Freshwater Research, 37, 399–420.
Chessman B.C. (2009) Climatic changes and 13-year trends
in stream macroinvertebrate assemblages in New
South Wales, Australia. Global Change Biology, 15, 2791–
2802.
Chessman B.C. (2013) Identifying species at risk from cli-
mate change: traits predict the drought vulnerability of
freshwater fishes. Biological Conservation, 160, 40–49.
Chester E.T. & Robson B.J. (2011) Drought refuges, spatial
scale and recolonisation by invertebrates in non-perennial
streams. Freshwater Biology, 56, 2094–2104.
Clarke H.G., Smith P.L. & Pitman A.J. (2011) Regional sig-
natures of future fire weather over eastern Australia from
global climate models. International Journal of Wildland
Fire, 20, 550–562.
Cooper S., Page H., Wiseman S., Klose K., Bennet D., Even
T. et al. (2014) Physicochemical and biological responses
of streams to wildfire severity in riparian zones. Freshwa-
ter Biology, doi:10.1111/fwb.12523, in press.
Costelloe J.F., Reid J.R.W., Pritchard J.C., Puckridge J.T.,
Bailey V.E. & Hudson P.J. (2010) Are alien fish disadvan-
taged by extremely variable flow regimes in arid-zone
rivers? Marine and Freshwater Research, 61, 857–863.
Cowell A.L., Matthews T.G. & Lind P.R. (2006) Effect of fire
on benthic algal assemblage structure and recolonization
in intermittent streams. Austral Ecology, 31, 696–707.
Crook D.A., Reich P., Bond N.R., McMaster D., Koehn J.D. &
Lake P.S. (2010) Using biological information to support
proactive strategies for managing freshwater fish during
drought. Marine and Freshwater Research, 61, 379–387.
CSIRO & Bureau of Meteorology (BOM) (2007) Climate
Change in Australia. CSIRO and Bureau of Meteorology,
Canberra, Australia.
CSIRO & Bureau of Meteorology (BOM) (2012) State of the Cli-
mate 2012. Australian Government, Canberra, Australia.
© 2014 John Wiley & Sons Ltd, Freshwater Biology, doi: 10.1111/fwb.12515
Extreme events and riverine ecosystems 15
CSIRO & Bureau of Meteorology (BOM) (2014) State of the Cli-
mate 2014. Australian Government, Canberra, Australia.
Dallas H. & Rivers-Moore N. (2012) Critical Thermal Max-
ima of aquatic macroinvertebrates: towards identifying
bioindicators of thermal alteration. Hydrobiologia, 679,
61–76.
Davies P., Cook B., Rutherford K. & Walshe T. (2004) Man-
aging high in-stream temperatures using riparian vegeta-
tion. River Management Technical Guideline No. 5. Land
& Water Australia, Canberra, Australia.
Donat M.G., Alexander L.V., Yang H., Durre I., Vose R.,
Dunn R.J.H. et al. (2013) Updated analyses of temperature
and precipitation extreme indices since the beginning of
the twentieth century: the HadEX2 dataset. Journal of Geo-
physical Research: Atmospheres, 118, 2098–2118.
Douglas M.M., Bunn S.E. & Davies P.M. (2005) River and
wetland food webs in Australia’s wet-dry tropics: general
principles and implications for management. Marine and
Freshwater Research, 56, 329–342.
Eliot M. (2012) Sea level variability influencing coastal
flooding in the Swan River region, Western Australia.
Continental Shelf Research, 33, 14–28.
Erskine W.D., Saynor M.J., Chalmers A. & Riley S.J. (2012)
Water, wind, wood, and trees: interactions, spatial varia-
tions, temporal dynamics, and their potential role in river
rehabilitation. Geographical Research, 50, 60–74.
Evans K. & Moliere D. (2010) Assessment of the significance
of extreme events in the Alligator Rivers Region – impact
of Cyclone Monica on Gulungul Creek catchment, Ranger
mine site and Nabarlek area. In: Eriss Research Summary
2008–2009. Supervising Scientist Report 201 (Eds D.R. Jones
& A.L. Webb ), pp. 179–184. Supervising Scientist, Dar-
win, Australia.
Frazer T.K., Notestein S.K., Jacoby C.A., Littlest C.J., Keller
S.R. & Swett R.A. (2006) Effects of storm-induced salinity
changes on submersed aquatic vegetation in Kings Bay,
Florida. Estuaries and Coasts, 29, 943–953.
Gallant A.J.E. & Gergis J. (2011) An experimental stream-
flow reconstruction for the River Murray, Australia,
1783–1988. Water Resources Research, 47, W00G04.
Gong W.P. & Shen J. (2009) Response of sediment dynam-
ics in the York River Estuary, USA to tropical cyclone
Isabel of 2003. Estuarine Coastal and Shelf Science, 84,
61–74.
Gunzburger M., Hughes W., Barichivich W. & Staiger J.
(2010) Hurricane storm surge and amphibian communi-
ties in coastal wetlands of northwestern Florida. Wetlands
Ecology and Management, 18, 651–663.
Harrison E., Dyer F., Wright D. & Levings C. (2014) Histori-
cal land-use influences the long-term stream turbidity
response to a wildfire. Environmental Management, 53,
393–400.
Hassall C. & Thompson D.J. (2008) The effects of environ-
mental warming on Odonata: a review. International Jour-
nal of Odonatology, 11, 131–153.
Hladyz S., Nielsen D.L., Suter P.J. & Krull E.S. (2012) Tem-
poral variations in organic carbon utilization by consum-
ers in a lowland river. River Research and Applications, 28,
513–528.
Ho S.S., Bond N.R. & Thompson R.M. (2013) Does seasonal
flooding give a native species an edge over a global inva-
der? Freshwater Biology, 58, 159–170.
Horner G.J., Baker P.J., Mac Nally R., Cunningham S.C.,
Thomson J.R. & Hamilton F. (2009) Mortality of develop-
ing floodplain forests subjected to a drying climate and
water extraction. Global Change Biology, 15, 2176–2186.
Horner G.J., Cunningham S.C., Thomson J.R., Baker P.J. &
Mac Nally R. (2012) Forest structure, flooding and graz-
ing predict understorey composition of floodplain forests
in southeastern Australia. Forest Ecology and Management,
286, 148–158.
Horrocks G.F.B., Cunningham S.C., O’Dowd D.J., Thomson
J.R. & Mac Nally R. (2012) Floodplain ants show a stron-
ger response to an extensive flood than to variations in
fallen-timber load. Austral Ecology, 37, 518–528.
Howes N.C., Fitzgerald D.M., Hughes Z.J., Georgiou I.Y.,
Kulp M.A., Miner M.D. et al. (2010) Hurricane-induced
failure of low salinity wetlands. Proceedings of the National
Academy of Sciences of the United States of America, 107,
14014–14019.
Hughes L. (2011) Climate change and Australia: key vulner-
able regions. Regional Environmental Change, 11, 189–195.
Humphries P. & Baldwin D.S. (2003) Drought and aquatic
ecosystems: an introduction. Freshwater Biology, 48, 1141–
1146.
Humphries P., King A.J. & Koehn J.D. (1999) Fish, flows
and flood plains: links between freshwater fishes and
their environment in the Murray-Darling river system,
Australia. Environmental Biology of Fishes, 56, 129–151.
IPCC (2012) Managing the risks of extreme events and
disasters to advance climate change adaptation. A special
report of Working Groups I and II of the Intergovern-
mental Panel on Climate Change, Cambridge University
Press, Cambridge, U.K., and New York, NY, U.S.A.
IPCC (2014) Climate change 2014: impacts, adaptation, and
vulnerability. Final draft of the Working Group II contri-
bution to the Fifth Assessment Report of the Intergovern-
mental Panel on Climate Change. http://www.ipcc.ch/
report/ar5/wg2/
Jardine T., Pusey B., Hamilton S., Pettit N., Davies P.,
Douglas M. et al. (2012) Fish mediate high food web con-
nectivity in the lower reaches of a tropical floodplain
river. Oecologia, 168, 829–838.
Jenkins K.M. & Boulton A.J. (2007) Detecting impacts and
setting restoration targets in arid-zone rivers: aquatic
micro-invertebrate responses to reduced floodplain inun-
dation. Journal of Applied Ecology, 44, 823–832.
Jones H.A. & Byrne M. (2010) The impact of catastrophic
channel change on freshwater mussels in the Hunter
River system, Australia: a conservation assessment. Aqua-
© 2014 John Wiley & Sons Ltd, Freshwater Biology, doi: 10.1111/fwb.12515
16 C. Leigh et al.
tic Conservation: Marine and Freshwater Ecosystems, 20, 18–
30.
Jones I. (Ed.) (2013) The Impact of Extreme Events on Freshwa-
ter Ecosystems. British Ecological Society, London, U.K..
Kang S.-R. & King S. (2012) Influence of salinity and prey
presence on the survival of aquatic macroinvertebrates of
a freshwater marsh. Aquatic Ecology, 46, 411–420.
Kennard M.J., Pusey B.J., Olden J.D., Mackay S.J., Stein J.L.
& Marsh N. (2010) Classification of natural flow regimes
in Australia to support environmental flow management.
Freshwater Biology, 55, 171–193.
King A.J., Tonkin Z. & Lieshcke J. (2012) Short-term effects
of a prolonged blackwater event on aquatic fauna in the
Murray River, Australia: considerations for future events.
Marine and Freshwater Research, 63, 576–586.
Lake P.S. (2000) Disturbance, patchiness, and diversity in
streams. Journal of the North American Benthological Society,
19, 573–592.
Lake P.S. (2003) Ecological effects of perturbation by drought
in flowing waters. Freshwater Biology, 48, 1161–1172.
Lake P.S. (2011) Drought and Aquatic Ecosystems: Effects and
Responses. Wiley-Blackwell, Chichester, U.K..
Lane P.N.J., Sheridan G.J. & Noske P.J. (2006) Changes in
sediment loads and discharge from small mountain
catchments following wildfire in south eastern Australia.
Journal of Hydrology, 331, 495–510.
Ledger M.E., Brown L.E., Edwards F.K., Milner A.M. &
Woodward G. (2013) Drought alters the structure and
functioning of complex food webs. Nature Climate Change,
3, 223–227.
Ledger M.E., Edwards F.K., Brown L.E., Milner A.M. &
Woodward G. (2011) Impact of simulated drought on
ecosystem biomass production: an experimental test in
stream mesocosms. Global Change Biology, 17, 2288–2297.
Ledger M.E., Harris R.M., Armitage P.D. & Milner A.M.
(2012) Climate change impacts on community resilience:
evidence from a drought disturbance experiment.
Advances in Ecological Research, 46, 211–258.
Leigh C., Burford M.A., Connolly R.M., Olley J.M., Saeck
E., Sheldon F. et al. (2013) Science to support manage-
ment of receiving waters in an event-driven ecosystem:
from land to river to sea. Water, 5, 780–797.
Leigh C., Sheldon F., Kingsford R.T. & Arthington A.H.
(2010) Sequential floods drive ‘booms’ and wetland per-
sistence in dryland rivers: a synthesis. Marine and Fresh-
water Research, 61, 896–908.
Lindenmayer D.B. & Likens G.E. (2010) The science and
application of ecological monitoring. Biological Conserva-
tion, 143, 1317–1328.
Lucas C., Hennessy K., Mills G. & Bathols J. (2007) Bushfire
weather in southeast Australia: recent trends and
projected climate change impacts. Consultancy Report to
the Climate Institute of Australia. Bushfire Cooperative
Research Centre and CSIRO Marine and Atmospheric
Research, Melbourne, Australia.
Lunt I.D., Jansen A. & Binns D.L. (2012) Effects of flood tim-
ing and livestock grazing on exotic annual plants in river-
ine floodplains. Journal of Applied Ecology, 49, 1131–1139.
Lyon J.P. & O’Connor J.P. (2008) Smoke on the water: can
riverine fish populations recover following a catastrophic
fire-related sediment slug? Austral Ecology, 33, 794–806.
Mackay S.M. & Robinson G. (1987) Effects of wildfire and
logging on streamwater chemistry and cation exports of
small forested catchments in southeastern New South
Wales, Australia. Hydrological Processes, 11, 359–384.
Mallin M.A., Posey M.H., Shank G.C., McIver M.R., Ensign
S.H. & Alphin T.D. (1999) Hurricane effects on water
quality and benthos in the Cape Fear watershed: natural
and anthropogenic impacts. Ecological Applications, 9, 350–
362.
Marshall J.C., Sheldon F., Thoms M. & Choy S. (2006) The
macroinvertebrate fauna of an Australian dryland river:
spatial and temporal patterns and environmental relation-
ships. Marine and Freshwater Research, 57, 61–74.
Matthews W.J., Surat E. & Hill L.G. (1982) Heat death of
the orangethroat darter Etheostoma spectabile (Percidae) in
a natural environment. Southwestern Naturalist, 27, 216–
217.
McCarthy B., Zukowski S., Whiterod N., Vilizzi L., Beesley
L. & King A. (2014) Hypoxic blackwater event severely
impacts Murray crayfish (Euastacus armatus) populations
in the Murray River, Australia. Austral Ecology, 39, 491–
500.
McGrath G.S., Sadler R., Fleming K., Tregoning P., Hinz C.
& Veneklaas E.J. (2012) Tropical cyclones and the ecohy-
drology of Australia’s recent continental-scale drought.
Geophysical Research Letters, 39, L03404.
Minshall G.W., Brock J.T. & Varley J.D. (1989) Wildfires
and Yellowstone’s stream ecosystems. BioScience, 39, 707–
716.
Morrongiello J.R., Beatty S.J., Bennett J.C., Crook D.A., Ike-
dife D.N.E.N., Kennard M.J. et al. (2011) Climate change
and its implications for Australia’s freshwater fish. Marine
and Freshwater Research, 62, 1082–1098.
Mosley L.M., Zammit B., Leyden E., Heneker T.M., Hipsey
M.R., Skinner D. et al. (2012) The impact of extreme low
flows on the water quality of the lower Murray River
and lakes. Water Resources Management, 26, 3923–3946.
Mouthon J. & Daufresne M. (2006) Effects of the 2003 heat-
wave and climatic warming on mollusc communities of
the Saone: a large lowland river and of its two main tri-
butaries (France). Global Change Biology 12, 441–449.
Murray B.B.R., Zeppel M.J.B., Hose G.C. & Eamus D. (2003)
Groundwater-dependent ecosystems in Australia: it’s
more than just water for rivers. Ecological Management and
Restoration, 4, 110–113.
Murray J.V., Stokes K.E. & Van Klinken R.D. (2011) Predict-
ing the potential distribution of a riparian invasive plant:
the effects of changing climate, flood regimes and land-
use patterns. Global Change Biology, 18, 1738–1753.
© 2014 John Wiley & Sons Ltd, Freshwater Biology, doi: 10.1111/fwb.12515
Extreme events and riverine ecosystems 17
National Climate Centre (NCC) (2010) Australia’s wettest
September on record but it is not enough to clear long-
term rainfall deficits. Special climate statement 22. Bureau
of Meteorology, Melbourne, Australia.
National Climate Centre, Bureau of Meteorology (NCC
BOM) (2011) Frequent heavy rain events in late 2010/
early 2011 lead to widespread flooding across eastern
Australia. Special climate statement 24. Bureau of Meteo-
rology, Melbourne, Australia.
National Climate Centre, Bureau of Meteorology (NCC
BOM) (2012) Australia’s wettest two-year period on
record; 2010–2011. Special climate statement 38. Bureau
of Meteorology, Melbourne, Australia.
Ning N.P., Gawne B., Cook R. & Nielsen D. (2013) Zoo-
plankton dynamics in response to the transition from
drought to flooding in four Murray–Darling Basin rivers
affected by differing levels of flow regulation. Hydrobiolo-
gia, 702, 45–62.
Nott J. (2011) A 6000 year tropical cyclone record from Wes-
tern Australia. Quaternary Science Reviews, 30, 713–722.
Paine R.T., Tegner M.J. & Johnson E.A. (1998) Compounded
perturbations yield ecological surprises. Ecosystems, 1,
535–545.
Papas P.J. (1998) Monitoring of fish and aquatic macroin-
vertebrates following the Caledonia Fire 1997/98 part I:
assessment of aquatic macroinvertebrate communities
and water quality January 1998. Report for the Depart-
ment of Natural Resources and Environment. Freshwater
Ecology, Parks, Flora and Fauna, Department of Natural
Resources and Environment, Heidelberg, Australia.
Peat M., Chester H. & Norris R. (2005) River ecosystem
response to bushfire disturbance: interaction with flow
regulation. Australian Forestry, 68, 153–161.
Perkins S.E. & Alexander L.V. (2013) On the measurement
of heat waves. Journal of Climate, 26, 4500–4517.
Pettit N. & Naiman R. (2005) Flood-deposited wood debris
and its contribution to heterogeneity and regeneration in
a semi-arid riparian landscape. Oecologia, 145, 434–444.
Pittock B., Abbs D., Suppiah R. & Jones R. (2006) Climatic
background to past and future floods in Australia.
Advances in Ecological Research, 39, 13–39.
Pohlman C.L., Goosem M. & Turton S.M. (2008) Effects of
Severe Tropical Cyclone Larry on rainforest vegetation
and understorey microclimate near a road, powerline and
stream. Austral Ecology, 33, 503–515.
Reich P. & Lake P.S. (2014) Extreme hydrological events
and the ecological restoration of flowing waters. Freshwa-
ter Biology, doi:10.1111/fwb.12508, in press.
Reid D.J., Quinn G.P., Lake P.S. & Reich P. (2008) Terres-
trial detritus supports the food webs in lowland intermit-
tent streams of south-eastern Australia: a stable isotope
study. Freshwater Biology, 53, 2036–2050.
Risbey J. (2011) Dangerous climate change and water
resources in Australia. Regional Environmental Change, 11,
197–203.
Robson B.J., Chester E.T., Allen M., Beatty S., Close P. &
Cook B. et al. (2013) Novel methods for managing fresh-
water refuges against climate change in southern Austra-
lia. National Climate Change Adaptation Research
Facility, Gold Coast, Australia.
Robson B.J., Chester E.T. & Austin C.M. (2011) Why life
history information matters: drought refuges and macro-
invertebrate persistence in non-perennial streams subject
to a drier climate. Marine and Freshwater Research, 62, 801–
810.
Rolls R.J., Leigh C. & Sheldon F. (2012) Mechanistic effects
of low-flow hydrology on riverine ecosystems: ecological
principles and consequences of alteration. Freshwater Sci-
ence, 31, 1163–1186.
Rose P., Metzeling L. & Catzikiris S. (2008) Can macroinver-
tebrate rapid bioassessment methods be used to assess
river health during drought in south eastern Australian
streams? Freshwater Biology, 53, 2626–2638.
Roshier D.A., Whetton P.H., Allan R.J. & Robertson A.I.
(2001) Distribution and persistence of temporary wetland
habitats in arid Australia in relation to climate. Austral
Ecology, 26, 371–384.
S�anchez-Arcilla A., Jim�enez J.A., Valdemoro H.I. & Gracia
V. (2008) Implications of climatic change on Spanish
Mediterranean low-lying coasts: the Ebro delta case. Jour-
nal of Coastal Research, 24, 306–316.
Saynor M. & Erskine W.D. (2008) Assessment of the signifi-
cance of extreme events in the Alligator Rivers Region:
impact of Cyclone Monica on riparian vegetation (Allo-
syncarpia ternata rainforest), in-channel large wood load-
ings, channel erosion and treethrow in the Ngarradj
Creek catchment. NOVA. The University of Newcastle’s
Digital Repository.
Schaefer J.F., Mickle P., Spaeth J., Kreiser B.K., Adams S.B.,
Matamoros W. et al. (2006) Effects of Hurricane Katrina
on the fish fauna of the Pascagoula River drainage. Water
Resources Research, 36, 62–68.
Sheldon F., Bunn S.E., Hughes J.M., Arthington A.H., Bal-
combe S.R. & Fellows C.S. (2010) Ecological roles and
threats to aquatic refugia in arid landscapes: dryland river
waterholes. Marine and Freshwater Research, 61, 885–895.
Sheldon F. & Fellows C.S. (2010) Water quality in two Aus-
tralian dryland rivers: spatial and temporal variability
and the role of flow. Marine and Freshwater Research, 61,
864–874.
Sheldon F., Marsh N. & Rolls R. (2012) Early warning, com-
pliance and diagnostic monitoring of ecological responses
to low flows. National Water Commission, Canberra,
Australia.
Smith M.D. (2011) An ecological perspective on extreme cli-
matic events: a synthetic definition and framework to
guide future research. Journal of Ecology, 99, 656–663.
Staben G.W. & Evans K.G. (2008) Estimates of tree canopy
loss as a result of Cyclone Monica, in the Magela Creek
catchment northern Australia. Austral Ecology, 33, 562–569.
© 2014 John Wiley & Sons Ltd, Freshwater Biology, doi: 10.1111/fwb.12515
18 C. Leigh et al.
Steffen W., Hughes L. & Karoly D. (Climate Commission)
(2013) The critical decade: extreme weather. Department
of Industry, Innovation, Climate Change, Science,
Research and Tertiary Education, Commonwealth of
Australia.
Steward A.L., Von Schiller D., Tockner K., Marshall J.C. &
Bunn S.E. (2012) When the river runs dry: human and
ecological values of dry riverbeds. Frontiers in Ecology and
the Environment, 10, 202–209.
Stewart B., Close P., Cook P. & Davies P. (2013a) Upper
thermal tolerances of key taxonomic groups of stream
invertebrates. Hydrobiologia, 718, 131–140.
Stewart R.I.A., Dossena M., Bohan D.A., Jeppesen E., Kor-
das R.L., Ledger M.E. et al. (2013b) Mesocosm experi-
ments as a tool for ecological climate-change research.
Advances in Ecological Research, 48, 71–181.
Thompson R.M., Beardall J., Beringer J., Grace M. & Sardina
P. (2013a) Mitigating impacts of climate change on stream
food webs. National Climate Change Adaptation
Research Facility, Gold Coast, Australia.
Thompson R.M., Beardall J., Beringer J., Grace M. & Sardina
P. (2013b) Means and extremes: building variability into
community-level climate change experiments. Ecology Let-
ters, 16, 799–806.
Thomson J.R., Bond N.R., Cunningham S.C., Metzeling L.,
Reich P., Thompson R.M. et al. (2012) The influences of
climatic variation and vegetation on stream biota: lessons
from the Big Dry in southeastern Australia. Global Change
Biology, 18, 1582–1596.
Timbal B. & Fawcett R. (2012) A historical perspective on
southeastern Australian rainfall since 1865 using the
instrumental record. Journal of Climate, 26, 1112–1129.
Townsend S.A. & Douglas M.M. (2000) The effect of three
fire regimes on stream water quality, water yield and
export coefficients in a tropical savanna (northern Austra-
lia). Journal of Hydrology, 229, 118–137.
Turner M.G., Baker W.L., Peterson C.J. & Peet R.K. (1998)
Factors influencing succession: lessons from large, infre-
quent natural disturbances. Ecosystems, 1, 511–523.
Verberk W.C.E.P. & Bilton D.T. (2013) Respiratory control
in aquatic insects dictates their vulnerability to global
warming. Biology Letters, 9, 20130473.
Verberk W.C.E.P., Van Noordwijk C.G.E. & Hildrew A.G.
(2013) Delivering on a promise: integrating species traits
to transform descriptive community ecology into a pre-
dictive science. Freshwater Science, 32, 531–547.
Verkaik I., Prat N., Rieradevall M., Reich P. & Lake P.S.
(2014) Effects of bushfire on macroinvertebrate communi-
ties in south-east Australian streams affected by a megad-
rought. Marine and Freshwater Research, 65, 359–369.
Watanabe K., Yaegashi S., Tomozawa H., Koshimura S. &
Omura T. (2014) Effects on river macroinvertebrate
communities of tsunami propagation after the 2011
Great East Japan Earthquake. Freshwater Biology, 59,
1474–1483.
Wedderburn S., Hammer M. & Bice C. (2012) Shifts in
small-bodied fish assemblages resulting from drought-
induced water level recession in terminating lakes of
the Murray-Darling Basin, Australia. Hydrobiologia, 691,
35–46.
Wernberg T., Smale D.A. & Thomsen M.S. (2012) A decade
of climate change experiments on marine organisms: pro-
cedures, patterns and problems. Global Change Biology, 18,
1491–1498.
White I., Wade A., Worthy M., Mueller N., Daniell T. &
Wasson R. (2006) The vulnerability of water supply catch-
ments to bushfires: impacts of the January 2003 wildfires
on the Australian Capital Territory. Australian Journal of
Water Resources, 10, 179–194.
Whitworth K.L., Baldwin D.S. & Kerr J.L. (2012) Drought,
floods and water quality: drivers of a severe hypoxic
blackwater event in a major river system (the southern
Murray–Darling Basin, Australia). Journal of Hydrology,
450-451, 190–198.
Woodward G., Brown L.E., Edwards F.K., Hudson L.N.,
Milner A.M., Reuman D.C. et al. (2012) Climate change
impacts in multispecies systems: drought alters food web
size structure in a field experiment. Philosophical Transac-
tions of the Royal Society B: Biological Sciences, 367, 2990–
2997.
Worthington Wilmer J., Murray L., Elkin C., Wilcox C., Nie-
jalke D. & Possingham H. (2011) Catastrophic floods may
pave the way for increased genetic diversity in endemic
artesian spring snail populations. PLoS ONE, 6, e28645.
(Manuscript accepted 3 November 2014)
© 2014 John Wiley & Sons Ltd, Freshwater Biology, doi: 10.1111/fwb.12515
Extreme events and riverine ecosystems 19