Mining protein dynamics from sets of crystal structures using “consensus structures
Distribution of neuropeptide FF-like immunoreactive structures in the lamprey central nervous system...
Transcript of Distribution of neuropeptide FF-like immunoreactive structures in the lamprey central nervous system...
Distribution of neuropeptide FF-like immunoreactivestructures in the lamprey central nervous systemand its relation to catecholaminergic neuronal structures
Manuel A. Pombal a,*, Jesus M. Lopez b, Marıa C. de Arriba a, Manuel Megıas a,Agustın Gonzalez b
aNeurolam Group, Department of Functional Biology and Health Sciences, Faculty of Biology, University of Vigo, 36310 Vigo, SpainbDepartment of Cell Biology, Faculty of Biology, University Complutense, 28040 Madrid, Spain
p e p t i d e s 2 7 ( 2 0 0 6 ) 1 0 5 4 – 1 0 7 2
a r t i c l e i n f o
Article history:
Received 1 April 2005
Accepted 22 June 2005
Published on line 17 February 2006
Keywords:
FMRFamide-related peptides
Neuropeptides
Immunohistochemistry
Hypothalamus
Agnathan
Evolution
a b s t r a c t
The neuropeptide FF (NPFF) is an octapeptide of the RFamide-related peptides (FaRPs) that
was primarily isolated from the bovine brain. Its distribution in the CNS has been reported in
several mammalian species, as well as in some amphibians. Therefore, in order to gain
insight in the evolution on the expression pattern of this neuropeptide in vertebrates, we
carried out an immunohistochemical study in the sea lamprey, Petromyzon marinus. The
distribution of NPFF-like-immunoreactive (NPFF-ir) structures in the lamprey brain is, in
general, comparable to that previously described in other vertebrate species. In lamprey,
most of the NPFF-ir cells were found in the hypothalamus, particularly in two large
populations, the bed nucleus of the tract of the postoptic commissure and the tuberomam-
millary area. Numerous NPFF-ir cells were also observed in the rostral rhombencephalon,
including a population in the dorsal isthmic gray and the reticular formation. Additional
labeled neurons were found inside the preoptic region, the parapineal vesicle, the periven-
tricular mesencephalic tegmentum, the descending trigeminal tract, the nucleus of the
solitary tract, as well as in the gray matter of the spinal cord. The NPFF-ir fibers were widely
distributed in the brain and the spinal cord, being, in general, more concentrated throughout
the basal plate. The presence of NPFF-ir fibers in the lamprey neurohypophysis suggests that
the involvement of NPFF-like substances in the hypothalamo-hypophyseal system had
emerged early during evolution.
# 2006 Elsevier Inc. All rights reserved.
avai lable at www.sc iencedi rec t .com
journal homepage: www.e lsev ier .com/ locate /pept ides
1. Introduction
The octapeptide neuropeptide FF (NPFF, FLFQPQRF-amide)
has been referred to as mammalian FMRF-NH2-like peptide,
F-8-F-amide, or morphine modulating neuropeptide. The
NPFF family consists of several different peptides: NPFF
and neuropeptide AF (NPAF), both originally isolated from
bovine brain by using antisera directed against the molluscan
* Corresponding author. Tel.: +34 986 812390; fax: +34 986 812556.E-mail address: [email protected] (M.A. Pombal).
0196-9781/$ – see front matter # 2006 Elsevier Inc. All rights reserveddoi:10.1016/j.peptides.2005.06.033
tetrapeptide FMRF-NH2 [88]; neuropeptide SF (NPSF), primarily
identified from the rat brain and spinal cord [89]; as well as
different longer peptides such as NPA-NPFF wich is the most
abundant in the rat spinal cord [9]; and SQA-NPFF, identified in
human neuroblastoma transfected with human proNPFF
transcript, and its homologue in mouse, SPA-NPFF [10]. Two
precursors (proNPFFA and proNPFFB) encoding peptides con-
taining the PQRF-amide sequence have been recently cloned
.
p e p t i d e s 2 7 ( 2 0 0 6 ) 1 0 5 4 – 1 0 7 2 1055
in mammals [58,81]. In a human cell line (neuroblastoma
SH-SY5Y), NPFF, NPAF, and SQA-NPFF are generated from
proNPFFA, whereas, NPFF, NPSF, and SPA-NPFF are identified
in the spinal cord of mice [10].
NPFF-like immunoreactivity (NPFF-ir) has been demon-
strated in the CNS of several mammalian species, including
rats ([33,34,42,45]; reviewed in [55,56]) and bovine brain [46],
as well as the spinal cords of mice, rats, guinea pigs and
humans [1,3,9,31,36,44]. Although NPFF has been detected in
the human plasma [72], its immunoreactivity seems to be
largely confined to the CNS, thus differing from a large
number of brain-gut peptides, which are found both in the
central and peripheral nervous system. In mammals, cell
bodies displaying NPFF-ir are generally restricted to a few
locations in the CNS: the central hypothalamus, supraoptic
and paraventricular nucleus, the nucleus of the solitary tract,
the spinal nucleus of the trigeminal tract, and different zones
of the dorsal horn in the spinal cord (see [55]). However,
numerous NPFF-ir fibers are widely distributed throughout
the mammalian CNS, with the highest innervations in the
posterior pituitary and dorsal horn of the spinal cord,
followed by the hypothalamus and the hindbrain [45,55].
Recently, a comparative study on the NPFF-ir structures in
the CNS of both anuran and urodele amphibians have also
been reported [18], and it represents the only report in
anamniote vertebrates. The presence of numerous NPFF-ir
fibers in the hypothalamo-neurohypophyseal system has
been observed in some of these studies [6,18,49].
In mammals, NPFF has been implicated in pain modulation
[1,23,29,88], opiate function [50,71,73], behavior [30], cardio-
vascular regulation [66], and neuroendocrine function
[2,47,48,49]; reviewed by Panula et al. [55]. The pharmacolo-
gical effects of NPFF and its analogs result from their
interactions with two G-protein-coupled receptors, NPFF1
and NPFF2. These receptors have been recently cloned and
characterized in rats and humans [8,20,25,38]. Available
information about the effects of NPFF on central neurotrans-
mitter systems is limited. According to pharmacological
experiments, NPFF has hyperalgesic effects [73,88] and it
elevates arterial blood pressure when injected intravenously
in rats [66]. In addition, both catecholamine-dependent and -
independent mechanisms appear to be involved in the
hypertensive effect of NPFF [66]. Tyrosine hydroxylase (TH)
is the rate-limiting enzyme in catecholamine biosynthesis,
and its immunohistochemical presence has been largely used
to identify catecholaminergic neurons (e.g., [60]). The presence
of NPFF-ir cells in many areas where TH immunoreactive (TH-
ir) neurons were previously found (see [59,60,62,84]), together
with the colocalization of these two substances in a
subpopulation of neurons in the nucleus of the solitary tract
of rats [32,42] promted us to check their presence in the same
cells in the lamprey’s CNS.
Among RFamide-related neuropeptides, antisera to the
molluscan cardio-excitatory tetrapeptide, FMRFamide, have
been used to identify FMRFamide-like immunoreactive mate-
rial in the CNS of many different invertebrate and vertebrate
species [5,24,26], including agnathans [12,15,19,28,54,85] and
many other fishes [13,16,61,77,78,87]. However, the presence
of NPFF has only been reported for some amphibian and
mammalian species [18,33,34,42,45,46,55,56].
The aim of this work was to study the distribution of NPFF-
ir structures in the brain of the adult sea lamprey, Petromyzon
marinus. Lampreys, together with hagfishes, are the only two
living representatives of the oldest lineage of vertebrates
(agnathans, jawless vertebrates). Interest in these groups of
fish is stimulated by their ancient lineage and the potential for
gaining insight into the evolution and distribution of neuro-
peptides, such as NPFF, in vertebrates.
2. Materials and methods
2.1. Animals
A total of 10 sexually mature adult sea lampreys (P. marinus;
seven males and three females) were used in the present
study. The animals were captured in the Mino River (north-
west part of Spain) during their upstream migration for
breeding, and obtained from a local supplier (Lampreas y
Angulas del Mino: Benıtez Fernandez, S.L.). The original
research reported here followed the guidelines on animal
care established by the Spanish Royal Decree 223/1988.
The animals were carried to the laboratory and processed
upon arrival. They were first deeply anesthetized in a 0.1%
solution of tricaine methanesulfonate (MS222; Sigma–Aldrich
Co., St. Louis, MO). Four animals were then perfused
transcardially with lamprey Ringer’s solution followed by
300 ml of 4% paraformaldehyde in 0.1 M phosphate buffer (PB;
pH 7.4), whereas, the other 6 animals were killed by
decapitation.
2.2. NPFF immunohistochemistry
The brain and rostral spinal cord was quickly dissected out
and fixed for 3–24 h at 4 8C in the same fixative solution. The
fixed tissues were then immersed in a solution of 30%
sucrose in PB at 4 8C until they sank, embedded in a solution
of 20% gelatin with 30% sucrose in PB, and immersed in a 4%
formaldehyde solution at 4 8C for 5 h. The gelatin blocks were
cut on a freezing microtome at 40 mm in the frontal or
sagittal plane and collected in PB. The tissue slices were then
rinsed twice in PB, treated with 1% H2O2 in PB for 15 min to
reduce endogenous peroxidase activity, and rinsed again
three times (10 min each) in PB. Sections were then
processed for immunohistochemistry by the peroxidase
anti-peroxidase (PAP) method [70]. Briefly, this included a
serial incubation of the sections in (1) a rabbit anti-NPFF
serum (kindly donated by Dr. H.-Y.T. Yang, NIH, Bethesda,
MD, USA) diluted 1:1000 in PB containing 2.5% Triton X-100,
for 48 h at 4 8C; (2) 0.1 M PB (3� 10 min); (3) swine anti-rabbit
serum (Nordic, Tilburg, The Netherlands), diluted 1:50 for
1 h; (4) 0.1 M PB (3� 10 min); (5) rabbit PAP complex
(Dakopatts, Denmark), diluted 1:500 for 90 min; (6) 0.1 M PB
(3� 10 min); (7) 0.5 mg/ml 3,30-diaminobenzidine (DAB;
Sigma, St. Louis) intensified with nickel, in 0.01% H2O2 in
distilled H2O; and (8) 0.05 M Tris–HCl buffer (TB, pH 7.6; 3�10 min). The secondary antiserum and the PAP complex were
diluted in PB containing 2.5% Triton X-100. Finally, the
sections were mounted (mounting medium, 0.25% gelatin in
TB) and, after drying overnight, added coverslips.
p e p t i d e s 2 7 ( 2 0 0 6 ) 1 0 5 4 – 1 0 7 21056
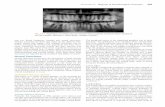
Fig. 1 – (A–O) Composite plate illustrating a set of representative transverse sections through the brain and rostralmost
spinal cord of Petromyzon marinus. The rostrocaudal level of each section is indicated in the schematic lateral view of the
brain as shown in (P). Photomicrographs of the left half of (A–O) show the overall distribution of NPFF-ir cells and fibers,
whereas, the specific names for neuronal groups or brain areas (drawn with dashed lines), major fiber tracts (drawn with
dotted lines), or individual large neurons (M3) are represented in the accompanied explanatory drawings (right part).
p e p t i d e s 2 7 ( 2 0 0 6 ) 1 0 5 4 – 1 0 7 2 1057
2.3. Double labeling for NPFF and TH
In four cases, a procedure based on immunohistofluores-
cence was used in a series of alternate sections as follows: (1)
incubation in a cocktail of mouse anti-TH (diluted 1:1000;
Incstar, Stillwater, MN, USA) and rabbit anti-NPFF (diluted
1:1000) in PB containing 2.5% Triton X-100, for 48 h at 4 8C; (2)
second incubation in a mixture of Alexa Fluor 594-conjugated
goat anti-rabbit (red fluorescence, diluted 1:300; Molecular
Probes, Eugene, OR, USA) and Alexa Fluor 488-conjugated goat
anti-mouse (green fluorescence, diluted 1:300; Molecular
Probes) for 90 min at room temperature; (3) after rinsing
three times in PB, the sections were mounted on glass slides
and coverslipped with Vectashield (Vector, Burlingame, CA,
USA); and (4) finally, the staining was studied and photo-
graphed with epifluorescent illumination by using the
appropriate filter combinations.
2.4. Evaluation and presentation of the results
The rabbit NPFF antiserum used in this study was previously
characterized and used in mammalian neuroanatomical
studies [27,46]. In addition, different sets of controls were
carried out in the amphibian brain [18] by preabsorption of the
antibody with bovine NPFF or a range of neuropeptides
structurally related and unrelated to the C-terminal part of
NPFF: FMRFamide, neuropeptide Y, adrenomedullin and
somatostatin, to test the specificity of the immunostaining.
The same antibody was used to detect the NPFF-ir structures
in the lamprey CNS following the immunohistochemical
procedures described in Sections 2.2 and 2.3.
The distribution of NPFF-like immunoreactive (NPFF-ir) cell
bodies and fibers in the brain of the adult sea lamprey is shown
Abbreviations: AB, anterobasal nucleus; aot, anterior octavomotor
cerebellar commissure; cc, central canal of the spinal cord; ch, o
region (gray matter) of the spinal cord; cs, central stratum (optic
dorsal column fibers; dh, dorsal horn of the spinal cord; dig, do
dmc, dorsomedial column of the spinal cord; dn, dorsal nucleus
tegmentum; dV, descending trigeminal tract; f, fasciculus retrofl
commissure; igl, internal granular layer of the olfactory bulb; II
trochlear nucleus; IX, glossopharyngeal motor nucleus; lc, latera
part of the lateral pallium; lpv, ventral part of the lateral pallium
mammillary region sensu lato; mlf, medial longitudinal fascicul
pallium; MPO, medial preoptic nucleus; mra, mesencephalic reti
mt, mesencephalic tegmentum; NH, neurohypophysis; nLLA, an
longitudinal fasciculus; NSM, nucleus of the stria medullaris; NT
of the tract of the postoptic commissure; nts, nucleus of the solit
area; on, optic nerve; os, optic stratum (optic tectum); ot, optic tec
paracommissural preoptic nucleus; PMg, magnocellular preopti
rhombencephalic reticular nucleus; ps, periventricular stratum
nucleus; PV, paraventricular nucleus; PVO, hypothalamic perive
mesencephalic recess; RM, retromammillary area; rm, mammil
rpro, preoptic recess; S, striatum; SCO, subcommissural organ;
intermedius ventralis; slh, sulcus limitans of His; SO, spino-occ
dorsal thalamus; TM, tuberomammillary nucleus; TN, tuberal nu
motor nucleus; vc, ventral column of the spinal cord; vd, dience
motor nucleus; vla, anterior lateral telencephalic ventricle; vlp, p
ventricle; vn, ventral nucleus of the octavolateral area; vr, rhom
vagal motor nucleus (rostral part). Scale bar represents 1 mm.
in Fig. 1. The drawings accompanying the pictures to show the
location of different areas, nuclei and major fiber tracts were
made by means of a camera lucida. Additional series of lamprey
stained brains from our collection were available for topo-
graphical purposes. In the present study, we followed the
nomenclature of Pombal and Puelles [65] for the forebrain, and
that of Pombal et al. [62,64] for the brainstem and spinal cord.
3. Results
The NPFF immunohistochemical procedure consistently
stained different populations of neurons in the lamprey
brain and spinal cord, and numerous NPFF-ir fibers were
widely distributed throughout the CNS. Most of the NPFF-ir
cells were observed in the hypothalamus, followed by the
isthmic region. The distribution and morphology of labeled
cells and fibers did not appear to vary between males and
females examined. Low power transverse pictures showing
the overall distribution of NPFF-ir cells and fibers in the
lamprey brain and spinal cord, as well as corresponding
detailed schematic drawings are shown in Fig. 1.
3.1. Distribution of NPFF-ir perikarya
3.1.1. Telencephalon
No NPFF-ir cell bodies were found in the olfactory bulb. The
most rostrally located group of NPFF-ir neurons was con-
stituted by scattered cells in the paraterminal preoptic nucleus
(Figs. 1A and 2A). The cells were spindle-shaped and
moderately immunostained and distributed throughout the
commissure of the lamina terminalis located in front of the
preoptic recess. More caudally, some weakly stained cells
tract; arn, anterior rhombencephalic reticular nucleus; cbc,
ptic chiasma; cpi, postinfundibular commissure; cr, cell
tectum); ctp, commissure of the tuberculum posterior; dcf,
rsal isthmal gray; dm, dorsomedial telencephalic neuropil;
of the octavolateral area; ds, dorsal sac; dt, diencephalic
exus; go, glomeruli olfactorii; HB, habenula; ibc, interbulbar
I, oculomotor nucleus; ip, interpeduncular nucleus; IV,
l column of the spinal cord; lp, lateral pallium; lpd, dorsal
; M3, third Muller cell; M5, nucleus M5 of Schober; MAM,
us; mn, medial nucleus of the octavolateral area; M, medial
cular area; mrn, medial rhombencephalic reticular nucleus;
terior lateral line nerve; nmlf, nucleus of the medial
P, nucleus of the tuberculum posterior; nTPOC, bed nucleus
ary tract; nV–nVIII, cranial nerves V–VIII; ola, octavolateral
tum; pc, posterior commissure; pch, choroidal plexus; PCP,
c nucleus; poc, postoptic commissure; prn, posterior
(optic tectum); PT, pretectum; PTP, paraterminal preoptic
ntricular organ; ri, infundibular recess; rlm, lateral
lary recess; rnp, neuroporic recess; rpo, postoptic recess;
SE, septum; SHL, subhippocampal lobe; siv, sulcus
ipital nucleus; ss, superficial stratum (optic tectum); TD,
cleus; to, optic tract; ts, torus semicircularis; V, trigeminal
phalic ventricle; VI, abducens motor nucleus; VII, facial
osterior lateral telencephalic ventricle; vm, mesencephalic
bencephalic ventricle; vt, impar telencephalic ventricle; Xr,
p e p t i d e s 2 7 ( 2 0 0 6 ) 1 0 5 4 – 1 0 7 21058
Fig. 1. (Continued).
were found in the compact band of closely packed cells that
constitute the magnocellular preoptic nucleus (Figs. 1B and
2B). In transverse sections, these cells were distributed
dorsally to the optic chiasm and were interspersed among
negative cell bodies. The NPFF-ir cells gave rise to two
processes: one apical process that coursed medially and
contacted the cerebrospinal fluid (CSF) in the preoptic recess;
and a basal process that coursed in the lateral neuropil
(Fig. 2B). A few NPFF-ir cells were also observed in the
paracommissural preoptic nucleus (Figs. 1C and 2C). These
were small piriform cells located just dorsal to the dorsal
postoptic commissure and were of CSF-contacting type
contacting with the impar telencephalic ventricle in the
medial part of the brain.
p e p t i d e s 2 7 ( 2 0 0 6 ) 1 0 5 4 – 1 0 7 2 1059
Fig. 2 – Photomicrographs showing NPFF-ir cells and fibers in transverse (A–C, F) and sagittal (D, E, G, H) sections through the
forebrain (A–G) and midbrain (H) of Petromyzon marinus. (A) Positive cells in the paraterminal preoptic nucleus (PTP) and
periventricular immunoreactive plexus in the septum (SE): rnp, neuroporic recess; (B) small weakly labeled cells (arrows) in
the magnocellular preoptic nucleus: rpro, preoptic recess; vt, impar telencephalic ventricle. (C) Many strong-labeled CSF-
contacting cells in the bed nucleus of the tract of the postoptic commissure (arrow) and few in the paracommissural
preoptic nucleus (arrowhead). (D) Numerous labeled cells in the bed nucleus of the tract of the postoptic commissure
(nTPOC), as well as in its rostral continuation, the anterobasal nucleus (AB): AH, adenohypophysis; ch, optic chiasma;
p e p t i d e s 2 7 ( 2 0 0 6 ) 1 0 5 4 – 1 0 7 21060
3.1.2. HypothalamusThe most conspicuous hypothalamic population of NPFF-ir
neurons was found in the anterobasal nucleus (Fig. 1B) and the
bed nucleus of the tract of the postoptic commissure (Fig. 1C
and D). This NPFF-ir population consists of numerous and
highly immunoreactive neurons located ventral and caudal to
the optic chiasm (Fig. 2C–E). These cells were round, piriform
or polygonal in shape, and possessed one thick and short
apical dendrite that reached the ventricular surface (Fig. 2C).
Immunopositive perikarya were intermingled among NPFF-
negative cells and arranged in three or four rows parallel to the
infundibular ventricular wall. The cell body of those NPFF-ir
cells located in the ependymal layer was small; whereas, the
most lateral ones showed larger somata (see the slightly
oblique parasagittal section shown in Fig. 3E). A few NPFF-ir
cells appeared displaced more caudally, thus entering the
limits of the hypothalamic periventricular organ. Numerous
processes originated from these cells coursed in several
directions, but mostly in the rostrocaudal plane innervating
the basal plate.
A second hypothalamic NPFF-ir group was detected in the
tuberomammillary area (Fig. 1E). The labeled cells occupied a
position ventral and slightly caudal to the bed nucleus of the
tract of the postoptic commissure. The cells were weakly
labeled and arranged parallel to the ventricle (Figs. 1E and 2F).
Moreover, some of the NPFF-ir cells contacted the CSF by
means of a club-shaped process (Fig. 2F).
3.1.3. DiencephalonWithin the diencephalon, only few NPFF-ir cells were observed
in the parapineal organ (Fig. 2G). These cells were weakly
labeled and had elongated bipolar cell bodies located in the
ventral wall of the parapineal vesicle. The pale cells gave rise
to more intensely stained processes with numerous and thick
varicosities that appears to innervate the neuropil of the
parapineal ganglion (Fig. 2G). Some labeled fibers were also
seen in the tela choroidea between the parapineal and the
habenula. The thalamus and the pretectum, as well as their
corresponding basal plate portions were devoid of NPFF-ir
cells in P. marinus (see Fig. 1E and F).
3.1.4. MesencephalonWithin the mesencephalon, one group of moderate NPFF-ir
cells was present in the subperiventricular tegmentum
(Fig. 1G). The neurons were small and possessed round cell
bodies with few and short processes (Fig. 2H). The NPFF-ir cells
were distributed dorsally to the dorsal rectus motor sub-
nucleus of the oculomotor nucleus, and thus can be
considered as belonging to the M5 nucleus of Schober, but
representing only a portion (ventral portion) of this retinopetal
nucleus.
Fig. 2. (Continued)
cpi, postinfundibular commissure; f, fasciculus retroflexus; NH,
bed nucleus of the tract of the postoptic commissure in a slightly
(thin arrow) and lateral (thick arrow) labeled cells. (F) Numerou
nucleus: ri, infundibular recess. (G) Weakly labeled cells (arrow
thick varicosities in the parapineal ganglion (G). (H) Immunorea
just dorsal to the third Muller cell (M3): vm, mesencephalic vent
200 mm (E, G, H).
3.1.5. RhombencephalonIn the dorsal part of the isthmic region, a group of numerous
NPFF-ir cells occupied a position from lateral to the trochlear
motor nucleus to the rostral portion of the trigeminal sensory
nucleus (Fig. 1H). The cells were scattered and weakly labeled
forming an arc throughout the isthmal gray, ventral to the so-
called cerebellar plate (Figs. 1H, 3A and B). The NPFF-ir cells
were multipolar, with fine, branching processes extending in
all directions, but forming a prominent plexus at the
periventricular side (Figs. 1H, 3A and B). The axons of these
cells appeared to fasciculate in the lateral part of the cell
group, forming a conspicuous and intense labeled fiber tract
that coursed ventromedially towards the midline (Fig. 1H and
I). From there, the axons could not be further followed because
of the dense network of labeled fibers and terminals within
which they were embedded.
More ventrally, some moderate to strong NPFF-ir cells were
located periventricularly, within the limits of the anterior
nucleus of the rhombencephalic reticular formation (Figs. 1H,
I, 3A and C, see also Fig. 4F). The cells were mostly located in
the caudal part of the nucleus. Most labeled reticular cells
were small, round or bipolar in shape, and grouped just below
the ependymal surface of the fourth ventricle (Fig. 3C).
Some NPFF-ir cell somata were primarily localized within
the limits of the descending trigeminal tract (Figs. 1J, 3D and E),
thus corresponding to the descending trigeminal tract
nucleus. The cells were only observed in the rostral one third
of this nucleus. The majority of these cells were small
multipolar neurons with short processes directed in several
directions (Fig. 3D and E).
In the caudal part of the rhombencephalic alar plate, some
strongly labeled neurons were found within the limits of the
nucleus of the solitary tract (Fig. 1M). Most of these cells were
grouped and laterally displaced, and located dorsal to the
descending trigeminal tract (Fig. 3F). The cells formed a
longitudinal band that can be clearly delineated in sagittal
sections (Fig. 3G) ending close to the level of the obex.
3.1.6. Spinal cordSome NPFF-ir cells were present in the dorsal aspect of the
spinal gray matter (Figs. 1O and 3H). These cells were mostly
monopolar, with a thick process that ramified dorsally and
originated the axon (Fig. 3H). Some bipolar cells were also
observed found on both sides of the midline, just ventral to the
lateral part of the dorsomedial column.
3.2. Distribution of NPFF-ir fibers
The NPFF-ir fibers were widely distributed in the adult sea
lamprey. Most of these fibers coursed longitudinally along
the laterobasal part of the brain, extending from the
neurohypophysis; rm, mammillary recess. (E) Detail of the
parasagittal section showing different size for the medial
s CSF-contacting cells (arrows) in the tuberomammillary
s) in the parapineal vesicle (V) and labeled fibers bearing
ctive cells in the mesencephalic periventricular nucleus,
ricle. Scale bar represents 100 mm (A–C, F), 500 mm (D), and
p e p t i d e s 2 7 ( 2 0 0 6 ) 1 0 5 4 – 1 0 7 2 1061
Fig. 3 – Photomicrographs showing NPFF-ir cells and fibers in transverse (A, C, D, F) and sagittal (B, E, G, H) sections through
the hindbrain of Petromyzon marinus. (A and B) Labeled cells in the dorsal isthmal gray nucleus: aot, anterior octavomotor
tract; arn, anterior rhombencephalic reticular nucleus; cbc, cerebellar commissure; vr, rhombencephalic ventricle. (C)
Immunoreactive cells (arrows) in the posterior part of the anterior rhombencephalic reticular nucleus, at the level of the
second isthmic Muller cell (I2). (D and E) Small labeled cells in the nucleus of the descending trigeminal tract: dV,
descending trigeminal tract. (F and G) Positive cells (arrows) in the nucleus of the solitary tract: dV, descending trigeminal
tract; slh, sulcus limitans of His; vn, ventral nucleus of the octavolateral area; Xr, vagal motor nucleus (rostral part). (H) Two
labeled cells and numerous fibers in the spinal cord. Scale bar represents 100 mm (A–G) and 25 mm (H).
p e p t i d e s 2 7 ( 2 0 0 6 ) 1 0 5 4 – 1 0 7 21062
Fig. 4 – Photomicrographs showing NPFF-ir fibers in transverse (A, C, D, G, H) and saggital (B, E, F) sections through the brain
and rostral spinal cord of the lamprey Petromyzon marinus. (A) Numerous labeled fibers in the neuropil of the striatum (S):
MPO, medial preoptic nucleus; vt, impar telencephalic ventricle. (B) Positive fibers in the ventral hypothalamus, including
the neurohypophysis (NH) and the postinfundibular commissure (cpi): rm, mammillary recess. (C) Detail of the
p e p t i d e s 2 7 ( 2 0 0 6 ) 1 0 5 4 – 1 0 7 2 1063
telencephalon up to the level of the spinal cord (Fig. 1). In
general, fibers showing NPFF immunoreactivity contained
bead-like varicosities along their trajectory, suggesting that
they form en passant synaptic contacts. The majority of
these fibers did not form discrete bundles, but were scattered
diffusely throughout diverse regions of the brain, with
particular accumulations in some nuclei as described herein.
3.2.1. TelencephalonIn the olfactory bulbs of the adult lampreys, only some
scattered NPFF-ir fibers were seen throughout of the internal
granular layer (Fig. 1A and B). These fibers did not enter in the
olfactory glomeruli, but a few were seen inside the olfactory
nerve. Some NPFF-ir fibers were also present in the pallium,
being more abundant in the ventral part of the lateral pallium
and the subhipoccampal lobe (Fig. 1B–D). In the subpallium,
labeled fibers were more numerous (Figs. 1A–C, 2A and 4A). In
the septal region, the labeled fibers accumulated in the vicinity
of the subventricular surface (Figs. 1A and 2A), whereas, in the
striatum region they were homogeneously distributed
throughout the periventricular neuropil (Figs. 1B, C and 4A).
3.2.2. HypothalamusA conspicuous fiber innervation was found throughout the
lamprey hypothalamus (Figs. 1B–F, 2B–F, 4B and C). These
fibers seem to be originated from the numerous NPFF-ir cells
located in this region. Most of these fibers coursed laterally,
and then turn either ventrally to enter the neurohypophysis
and the postinfundibular commissure, or caudally to inner-
vate the brainstem and the spinal cord. The neurohypophysis
was richly innervated from rostral to caudal with the
immunoreactive fibers showing numerous thick varicosities
(Figs. 2D and 4B). In sagittal sections, part of the labeled fibers
that innervate the neurohypophysis could be followed to their
place of origin in the bed nucleus of the tract of the postoptic
commissure. The postinfundibular commissure was also richly
innervated with the largest accumulation of labeled fibers in
its superficial half portion (Figs. 1F, 2D, 4B and C). Other fibers
coursed rostrally to innervate the preoptic area as well as the
subpallial and pallial regions, and the olfactory bulbs. Finally,
some fibers coursed dorsally to innervate partially the dorsal
diencephalon and mesencephalon (see Fig. 1D–G). These
fibers were relatively thick in the vicinity of the cell body
and became thinner along their trajectory. In addition, some
labeled fibers were also detected in the optic nerves (Fig. 1B).
3.2.3. DiencephalonIn the diencephalon, the alar plate of both ventral and dorsal
thalamus were poorly innervated, however some NPFF-ir
postinfundibular commissure (cpi): rm, mammillary recess. (D)
ventricle. (E) Distribution of labeled fibers in the superficial (ss),
optic tectum: vm, mesencephalic ventricle. (F) Labeled fibers in
with different intensities of immunoreaction in the interpedunc
posterior part of the anterior rhombencephalic reticular nucleus
ventricle. (G) NPFF-ir fibers concentrate in the periventricular ne
motoneurons of the trigeminal motor nucleus (V): mrn, medial
His; vr, rhombencephalic ventricle. (H) Distribution of labeled fi
columns, as well as in the dorsal horn (dh) and the cell region (c
100 mm (A–E, G, H) and 500 mm (F).
fibers accumulated in the habenula and its vicinity (Figs. 1E
and 4D), as well as in the ganglion of the parapineal organ
(Fig. 2G). In this ganglion, numerous labeled fibers with thick
varicosities were evenly distributed (Fig. 2G). The pretectal alar
plate was homogeneously innervated by NPFF-ir fibers, with
the exception of the optic tract (that is almost devoid of labeled
fibers throughout its rostrocaudal extent), and the subcom-
missural organ, where only few fibers could be seen (Fig. 1F).
Ventrally, some fibers crossed in the commissure of the
tuberculum posterior and the subpretectal commissure,
whereas, dorsally the fibers crossed in the posterior commis-
sure (Fig. 1F).
3.2.4. MesencephalonA moderate number of NPFF-ir fibers were also present in the
mesencephalic tegmentum as well as in the dorsal optic
tectum (Fig. 1G). In the tegmentum, the labeled fibers were
evenly distributed in the medial and lateral portions, while the
subventricular region where most cell bodies were located was
poorly innervated (Fig. 1G). Some fibers were also seen inside
the oculomotor nucleus intermingled with the motoneurons
(Figs. 1G and 2H). In the optic tectum, the NPFF-ir fibers
showed a precise fiber arrangement that is conserved
throughout its rostrocaudal extent. The labeled fibers were
more abundant in the periventricular portion, including the
periventricular and central strata, than in the superficial
portion, including the optic and superficial strata (Figs. 1G and
4E). However, the dorsomedial border of the optic tectum
displayed the largest accumulation of immunoreactive fibers
(Fig. 1G); some of these fibers crossed the midline at the
caudally located posttectal commissure.
3.2.5. RhombencephalonThe isthmic region, together with the hypothalamus, showed
the highest concentration of NPFF-ir fibers in the lamprey
brain. One of the major accumulations of immunoreactive
fibers within the isthmic region occurred in the dorsal part,
just at the level of the labeled cells (Figs. 1H, I, 3A and B). A
particular dense network of thin fibers was present in the
ventromedial neuropil of the interpeduncular nucleus
(Figs. 1G–I, 3C and 4F). Rostrocaudally, the interpeduncular
nucleus spread from the oculomotor nucleus to the level of the
first half of the trigeminal motor nucleus. In our material,
different densities for NPFF-ir fibers were detected in
consecutive rostrocaudal regions of the interpeduncular
neuropil, with the highest concentration at both sides of the
midline (see Figs. 1G–I and 3C). More laterally, several portions
particularly well delineated were undoubtedly distinguishable
in sagittal sections (Fig. 4F). It includes two parts with very few
Labeled fibers in the habenula (HB): vd, diencephalic
optic (os), central (cs), and periventricular (ps) strata of the
the caudal mesencephalon and rostral rhombencephalon,
ular neuropil (ip). Some labeled cells are also present in the
(arn): vm, mesencephalic ventricle; vr, rhombencephalic
uropil, whereas, few are located among the large
rhombencephalic reticular nucleus; slh, sulcus limitans of
bers in the dorsomedial (dmc), lateral (lc), and ventral (vc)
r) of the spinal cord: cc, central canal. Scale bar represents
p e p t i d e s 2 7 ( 2 0 0 6 ) 1 0 5 4 – 1 0 7 21064
Fig. 5 – Photomicrographs showing NPFF-ir (in red) and TH-ir (in green) cells and fibers in sagittal (A, E–G) and transverse
(B–D, H) sections through the forebrain (A–G) and midbrain (H) of Petromyzon marinus. (A) General view of the lamprey
hypothalamus: AH, adenohypophysis; ch, optic chiasma; cpi, postinfundibular commissure; NH, neurohypophysis; rm,
mammillary recess. (B) NPFF-ir (in red) and TH-ir (in green) CSF-contacting cells in the bed nucleus of the tract of the
postoptic commissure (arrow) and the paracommissural preoptic nucleus (arrowhead). (C) NPFF-ir (in red) and TH-ir
p e p t i d e s 2 7 ( 2 0 0 6 ) 1 0 5 4 – 1 0 7 2 1065
labeled fibers separated by a smaller portion moderately
innervated, and a caudal part richly innervated (Fig. 4F).
Another important aspect of the NPFF-ir fibers in the lamprey
isthmus is the presence of a thin but well delineated tract of
labeled fibers that originated from the NPFF-ir cells located in
the dorsal isthmic gray. These fibers join together in the
dorsolateral aspect of the nucleus, just in front of the
emergence of the sensory and motor trigeminal roots. Then,
the fibers coursed ventromedially to reach and cross the
ventral midline through a discrete commissure located above
the heavily immunoreactive neuropil of the interpeduncular
nucleus (Fig. 1H and I). Thereafter, it was not possible to
determine the location of the target areas or nuclei of these
fibers, but it seems that they turn to constitute a diffuse
longitudinal tract together with other descending labeled
fibers from the hypothalamus.
In the rhombencephalon proper, there are several aspects
of the innervation by NPFF-ir fibers that deserve a comment.
Most of the labeled fibers were distributed throughout the
whole basal plate, whereas, labeled fibers in the alar plate
were very scarce (Fig. 1I–N). In the alar plate, a fine network of
thin immunoreactive fibers extended over all octavolateral
nuclei, with some more fibers at the border of the insertion of
the tela choroidea that covers the fourth ventricle, as well as
inside the descending trigeminal tract (Figs. 1I–N, 3D and E). In
the basal plate, the labeled fibers extended over most of the
tegmentum, but they were more abundant laterally at the
border with the descending trigeminal tract (Fig. 1I–N). Of
interest is the remarkable labeled periventricular dense fiber
plexus of the rhombencephalic branchiomotor column,
including the trigeminal, the facial, the glossopharingeal
and the vagal motor nuclei (Figs. 1I–N, 3B, C, F and 4G).
3.2.6. Spinal cordIn the spinal cord, labeled fibers were found in the lateral
columns, with the highest density at the border with the
dorsal columns, and, to a lesser extent, in the ventral and
dorsal columns (Figs. 1O, 3H and 4H). Part of them constituted
a caudal continuation of the longitudinal tracts in the
rhombencephalon, whereas, others originated from local
positive cells. In the dorsal column, labeled fibers were more
numerous ventromedially. Some thin and varicose fibers were
also seen between the motoneurons, inside the spinal motor
column (Fig. 4H).
3.3. Double NPFF and TH immunohistochemistry
The pattern observed for the distribution of NPFF immunor-
eactivity in the sea lamprey brain pointed to the possibility
of extensive codistribution with catecholaminergic cells
[62,59,60,84]. Both NPFF-ir and TH-ir cells were found in the
(in green) cells in the caudal part of the bed nucleus of the tract
(E) Part of the hypothalamus showing NPFF-ir cells (in red) in its
partial overlapping in the caudal part of the bed nucleus of the
commissure; rm, mammillary recess. (F) Detail of the square in
commissure; PVO, hypothalamic periventricular organ. (G) NPFF
neurohypophysis (NH). (H) NPFF-ir (in red) and TH-ir (in green) f
midline (arrow). Scale bar represents 500 mm (A), 100 mm (B, C,
commissure of the paraterminal preoptic nucleus (nucleus of
the anterior commissure [62]), the small cells of the magno-
cellular preoptic nucleus, the paracommissural preoptic
nucleus (a part of the nucleus of the postoptic commissure
in Pierre et al. [59,60], and Pombal and Puelles [65]), the rostral
part of the bed nucleus of the tract of the postoptic
commissure (another part of the nucleus of the postoptic
commissure in Pierre et al. [59,60], and Pombal and Puelles
[65]), the caudal hypothalamus, the rostral rhombencephalon,
the nucleus of the solitary tract, and the spinal cord. However,
in double fluorescent immunolabeled sections it became clear
that there is a high degree of codistribution of these two
substances, but colocalization in the same neuron was absent.
Both types of immunoreactive cells were clearly interspersed
in the preoptic area, including the commissure of the
paraterminal preoptic nucleus, the small cells of the magno-
cellular preoptic nucleus, and the paracommissural preoptic
nucleus (Fig. 5A and B), as well as in the rostral part of the bed
nucleus of the tract of the postoptic commissure (Fig. 5B). More
caudally in the hypothalamus, some cells of the caudal part of
the bed nucleus of the tract of the postoptic commissure
(Fig. 5C and D) and the tuberommamillary nucleus (Fig. 5E and
F) codistributed with catecholaminergic cells rostrally
migrated, whereas, in the hypothalamic periventricular organ,
and the mammillary and retromammillary regions (Fig. 5A, E
and F) only TH-ir cells were observed. Unfortunately, an
assessment of colocalization of NPFF and TH-ir cells in either
the isthmic region, the nucleus of the solitary tract, and the
spinal cord was not possible because of the absence of
immunolabeling with the anti-TH antiserum used in this
study. In addition, there was a high overlapping of labeled
NPFF and TH-ir fibers throughout the lamprey brain and spinal
cord, such as in the striatum region, the neurohypophysis
(Fig. 5A and G), the postinfundibular commissure (Fig. 5A and
E), or the optic tectum (Fig. 5H), but colocalization of both
substances was not observed in labeled fibers.
4. Discussion
The aim of this study was to provide a detailed description of
the topographical localization of NPFF-ir structures in the
central nervous system of lampreys, which actually repre-
sents the first study on the distribution pattern of this
neuropeptide in fishes. The study was performed in adult P.
marinus by using an antiserum against bovine NPFF. Our
results in the CNS of upstream-migrating adult P. marinus
show the presence of NPFF-ir cells mainly in the hypothala-
mus and the isthmic region. The comparative analysis of the
distribution of NPFF-ir structures reported herein with those
previously reported in agnathans for FMRFamide [19,54],
of the postoptic commissure. (D) Detail of the square in C.
rostral part and TH-ir cells (in green) in its caudal part, with
tract of the postoptic commissure: cpi, postinfundibular
(E). nTPOC, bed nucleus of the tract of the postoptic
-ir (in red) and TH-ir (in green) fibers in the
ibers in the optic tectum, with some crossing at the caudal
G, H), 50 mm (D), and 200 mm (E).
p e p t i d e s 2 7 ( 2 0 0 6 ) 1 0 5 4 – 1 0 7 21066
Table 1 – Comparative analysis of the cell populations immunoreactive for NPFF, FMRF, and NPY in lampreys
PTP SHL PMg AB nTPOC TM Th mt dig arn mrn ndV ola nts Spinal cord
NPFF (1) + � + + + + � + + + � + � + +
FMRF (2) � � � � + � � � + � � � � � �FMRF (3) � � � � + � � � � � � � � � �NPY (4) � + + + + � + + � + + � + � +
NPY (5) * + � + + � + * * + + * + * +
The presence (+) or absence (�) of immunoreactivity (ir) is indicated in the different populations obtained from the results in (1) Petromyzon
marinus, present study, (2) Ichthyomyzon unicuspis [19], (3) Lampetra japonica [53], (4) Lethenteron japonica [17], and (5) Lampetra fluviatilis and/or
Ichthyomyzon unicuspis [7,11,56,64,77]. The asterisk (*) indicates populations that were not studied. Abbreviations: AB, anterobasal nucleus; arn,
anterior rhombencephalic reticular nucleus; dig, dorsal isthmal gray; mrn, medial rhombencephalic reticular nucleus; mt, mesencephalic
tegmentum; ndV, nucleus of the descending trigeminal tract; nTPOC, bed nucleus of the tract of the postoptic commissure; nts, nucleus of the
solitary tract; ola, octavolateral area; PMg, magnocellular preoptic nucleus; PTP, paraterminal preoptic nucleus; SHL, subhippocampal lobe; Th,
thalamus; TM, tuberomammillary nucleus.
and/or neuropeptide Y (NPY) [11,17,65,79,88] showed marked
differences (see Table 1 and Section 4.2), suggesting that a
NPFF-like peptide exists in lampreys. Finally, the relationship
of the distribution of NPFF-ir to catecholaminergic (TH-ir)
cells was analyzed by using a double immunohistochemistry
technique.
4.1. Comparison of NPFF-ir structures in the lampreybrain with previous results in amphibians and mammals
In the following paragraphs, we compare our findings on the
distribution of NPFF-immunoreactivity in the lamprey CNS
with those previously reported on several mammalian species
[33,34,42,45,46,55,56], as well as in some amphibians (Rana
perezi, Xenopus laevis, and Pleurodeles waltl; [18].
4.1.1. TelencephalonIn this research, no NPFF-ir cells were found in the olfactory
bulbs or the telencephalon of the adult sea lamprey. Labeled
cells in these two brain subdivisions have only been detected
in amphibians [18]. Concerning the NPFF-ir fibers, the
distribution pattern in lampreys with scattered fibers in the
olfactory bulbs and pallial subdivisions, and numerous fibers
in the subpallium, including the septum and the striatum, is
more similar to that reported in amphibians [18] than that
found in mammals, where the cortical regions and the basal
ganglia are almost free of labeling or poorly innervated
[33,46,55]. More studies are needed covering a more diverse
range of vertebrates before determining whether these
differences represent an evolutionary change between ana-
mniote and amniote vertebrates.
Finally, very few NPFF-ir fibers were present in the optic
nerve. In this nerve, occasional fibers were also immunoreac-
tive for either GnRH or FMRFamide [19]. A possibility raised by
these authors, is that these fibers could be homologous with
the retinopetal component of the terminal nerve system that
has been characterized in many other vertebrates (e.g., [51,86]).
Curiously enough, there is the presence of NPFF-ir cells in the
ganglion of the terminal nerve in both Carassius auratus [40],
and Pleurodeles waltl [18] with the same antibody used in this
study. In addition, a NPFF-related gene recently cloned in
zebrafish (Danio rerio) is selectively expressed only in neurons of
the terminal nerve [53]. However, based on experimental
material regarding the organization of the retinal projections
and immunohistochemistry for those substances reported in
other species, the existence of a terminal nerve in lampreys is
still very controversial (see [19,82]).
4.1.2. HypothalamusAs in mammals and amphibians, this study reveals that most
of the NPFF-ir cells in the lamprey brain are located in the
hypothalamus, including distinct subdivisions of the preoptic,
tuberal, and mammilar regions. In amphibians there are NPFF-
ir cells in the suprachiasmatic nucleus, the retrochiasmatic
nucleus, and the anterodorsal portion of the infundibulum of
anurans, whereas, in urodeles they are located in the anterior
preoptic area, the suprachiasmatic nucleus, and the dorsal
aspect of infundibular hypothalamus [18]. In mammals most
NPFF-ir cells were initially located between dorsomedial and
ventromedial hypothalamic nucleus (i.e., in the intermedio-
medial nucleus; see [55]); but some were also reported in the
supraoptic and paraventricular nuclei [6,31]. However, it is
now known that the NPFF immunoreactivity of the inter-
mediomedial nucleus in mammals is due to other peptide
belonging to the NPVF family of peptides [43,76]. Although no
definitive comparisons between species concerning labeled
nuclei can be made, some similarities as well as differences
appear to exist. As an example, the neurosecretory neurons of
the paraventricular nucleus of rats have been shown to
contain both NPFF and vassopressin and to project to the
neurohypophysis [49], where both substances colocalize in
nerve terminals as visualized by post-embedding immunoe-
lectron microscopy [41]. However, the paraventricular nucleus
of lampreys that contains vasotocin (the lamprey counterpart
of vassopressin; [65]) and projects to the neurohypophysis (see
[63,64]) was not immunoreactive for NPFF. There are numer-
ous vasotocin immunolabeled cells projecting to the neuro-
hypophysis in the magnocellular preoptic nucleus [22,65], and
only the small NPFF-ir cells of this nucleus could contain
vasotocin, which seems highly unlikely. However, the NPFF-ir
cells in the dorsal infundibular portion of the amphibian
hypothalamus [18], and the intermediomedial nucleus of
mammals [33,34,35,37,42,56], could be considered equivalent,
at least in part, to the bed nucleus of the tract of the postoptic
commissure and/or the tuberomammillary nucleus of lam-
preys. Some correspondence could be established between the
labeled cells located in the preoptic and supraoptic areas of
lampreys, amphibians, and mammals.
p e p t i d e s 2 7 ( 2 0 0 6 ) 1 0 5 4 – 1 0 7 2 1067
Concerning the NPFF innervation, the hypothalamus of
amphibians and mammals are richly innervated by labeled
fibers [18,41,49]. The lateral walls of the lamprey hypothalamus
are also innervated by a conspicuous fiber plexus, which
partially enters the neurohypophysis. In the hypophysis of
amphibians, the intermediate lobe is primarily innervated by
NPFF-ir fibers [18], whereas in rat labeled fibers enter the neural
lobe (median eminence [6,31,41,49]. In lampreys, the anterior
neurohypophysis has been compared to the mammalian
median eminence, whereas, the posterior neurohypophysis
would correspond to the neural lobe (see [74]. Although not
evenly distributed, our results demonstrated that both
parts are highly innervated by varicose fibers; therefore,
this peptide may be involved in hypothalamic regulation
of pituitary function in lampreys as previously shown in
mammals [47,49].
4.1.3. DiencephalonIn the sea lamprey diencephalon, NPFF-ir cells were only
present in the parapineal vesicle with a rich fiber innervation
in its ganglion. This is the first time that some NPFF
immunoreactivity is reported in the epiphysis of vertebrates.
The bipolar shape of the labeled cells in the ventral wall of the
parapineal vesicle suggests that the cells correspond to
photoreceptors. These cells give rise to nerve terminals in
the parapineal ganglion, as well as to some fibers running in
the parapineal tract, and thus appear to form part of both
local and external circuits. In a previous study cells immu-
noreactive for choline acetyltransferase (ChAT-ir) were
reported in the ventrocaudal region of the parapineal vesicle
[90]. Although the distribution of both NPFF-ir and the
ventrocaudal ChAT-ir cell group is similar, they probably
represent separated populations on the basis of the different
thickness and shape of cells; however, this should be
confirmed by using double labeling. Apart from the labeled
cells of the lamprey parapineal, NPFF-ir cells have been also
found in the central nucleus of the dorsal thalamus of
Xenopus [18].
4.1.4. MesencephalonIn this study of the lamprey, we found a single cell population
of NPFF-ir neurons in the ventromedial mesencephalic
tegmentum. It is not clear as to whether or not the labeled
cells belong to the M5 nucleus of Schober, where some of the
retinopetal cells are located in these animals (see [80] for a
review). If it is assumed that the NPFF-ir cells correspond with
the retinopetal cells, then the presence of some labeled fibers
in the optic nerves could represent the axons of these cells on
the way to the retina. A similar population was previously
reported in the mesencephalon of Xenopus, but located
laterally [18]; however, in amphibians there are no retinopetal
cells located in the mesencephalon (see [75] for a review).
Therefore, the labeled cells in the lamprey could correspond to
or be homologous of those reported in Xenopus. In lamprey,
tachykinin-ir cells [4] were also reported in a similar location.
The presence of numerous NPFF-ir fibers in either the
mesencephalic tegmentun and optic tectum of the lamprey
brain is more similar to what was reported in amphibians [18]
than in mammals, where the NPFF innervation was scarce or
absent [33,55].
4.1.5. RhombencephalonThe isthmus of lampreys possesses numerous NPFF-ir cells
located in two populations, one located in the dorsal isthmal
gray and other belonging to the reticular formation. However,
no labeled cells were previously found in the isthmus of the
other species of vertebrates studied. Of interest is the
existence of abundant labeled fibers originated from the
positive isthmic cells of lampreys that appears to innervate
the ventromedial interpeduncular neuropil, as well as to joint
the rostrocaudal fiber tracts that run up to the spinal cord. An
extremely intense longitudinal fiber tract from the level of the
isthmus to the spinal cord was shown in urodele amphibians
[18]. The origin of this tract is unclear. One possibility is that
the cells are located in the dorsal portion of the isthmus, but
cannot be distinguished because of the high concentration of
labeled fibers in such region.
Many NPFF-ir cells were present in the rhombencephalic
reticular formation of the lamprey, but restricted to the
anterior rhombencephalic reticular nucleus. Immunolabeled
reticular cells have been shown also in the median and inferior
reticular formation of anurans, but not in urodele amphibians
[18]. Conversely, in the reticular formation of the mammals
studied so far, no labeled NPFF-ir cells were found.
The immunopositive neurons located in the trigeminal
sensory nucleus of lampreys are not evenly distributed
throughout the nucleus but occupy the rostral one third,
where sensory information carried by fibers of the five
branches of the trigeminal nerve (i.e., the ophthalmic, apical,
basilar, suborbital, and mandibular) appears to be processed
[39]. However, the location of the NPFF labeled cells, i.e., the
ventral part of the descending trigeminal tract, better
corresponds with the distribution area of fibers from the
ophthalmic branch [39]. In rats, a strong signal for NPFF mRNA
was found in the spinal nucleus of the trigeminal nerve by in
situ hybridization [81].
The motor neurons of the branchial nerves themselves also
appear to receive an NPFF innervation. The branchiomotor
column of lampreys (including the trigeminal, the facial, the
glossopharingeal and the vagal motor nuclei) consists of a
more or less continuous band of neurons along the rhom-
bencephalon (see [64]). The density of NPFF positive fibers is
relatively low around the neuronal somata, but moderate on
the ventrolateral aspect of the cell column and high on its
periventricular margin, where the main dendrites of the
motoneurons extend. In mammals, NPFF has been implicated
in sensory systems, most notably pain and morphine
analgesia [88]. The location of the immunopositive cell bodies
in the trigeminal sensory nucleus and the presence of a
terminal arborization around the branchiomotor column
suggest the NPFF plays a role in the sensory-motor control
of the branchial apparatus, even though many details of this
interaction remain obscure.
The presence of NPFF-ir cells in the nucleus of the solitary
tract is a common feature of all vertebrates studied so far,
including lampreys [18,33,56]. In addition, in rats some of the
neurons, which are positive for NPFF are also positive for
tyrosine hydroxylase [32,42]; see also [55]. Although some
neurons have been shown to be immunoreactive for dopa-
mine in a location considered in lampreys to be homologous to
the nucleus of the solitary tract [62], the anti-TH monoclonal
p e p t i d e s 2 7 ( 2 0 0 6 ) 1 0 5 4 – 1 0 7 21068
antibody used in this study did not label any cells in that
position (see Section 4.3). Therefore, it cannot be confirmed
whether or not both substances coexist in the same cells of
this population.
4.1.6. Spinal cordSome NPFF-ir cells have been detected in the central gray
matter of the lamprey spinal cord. Weakly labeled cells in a
similar location were found in amphibians [18], which have
been suggested to be comparable to those identified in the
spinal cord of the rats [36]. The innervation of the spinal cord
by NPFF-ir fibers is more conspicuous in the dorsal part,
particularly in the dorsal horn, in all species studied so far
[18,21,36,45]. Although experiments with tracing techniques
suggest that NPFF immunoreactivity in the spinal cord of rats
is of intrinsic origin [36], some contribution from a descending
tract has been suggested [21,45]. Although there are some
labeled cells in the spinal cord of lampreys with their
corresponding processes, most of the fibers innervating the
spinal cord constituted a caudal continuation of the long-
itudinal tracts found in the rhombencephalon. This is
consistent with results obtained in amphibians [18], where
the innervation of the spinal cord by descending fibers forming
a strong immunoreactive tract is unequivocally distinguished
in urodeles.
4.2. Comparison with other RFamide and RYamide-relatedpeptides reported in the lamprey brain
The distribution pattern of NPFF-ir cell bodies and fibers in the
lamprey brain shows many similarities to those of other
neuropeptides (for review see [52]). Furthermore, some
polyclonal antisera to FMRF-amide (a member of the RFa-
mide-related peptides: FaRPs) or NPFF have been found to
weakly cross-react with neuropeptide Y (NPY, a member of the
RYamide-related peptides) [46,67]. However, a comparative
analysis of the distribution of these neuropeptides to that of
NPFF in lampreys showed marked differences (see Table 1)
pointing to a lack of cross-reactivity of the antiserum used in
this study with those neuropeptides.
In this section, we discuss our results concerning the
labeling found in agnathans with antisera against FMRFamide
and NPY noting that cross-reactivity of our polyclonal anti-
body for NPFF with these peptides could also be expected.
Previous studies have reported the distribution of FMRFamide-
like immunoreactivity (FMRF-ir) using different antisera in the
agnathan brain, including three hagfish species: Eptatretus
burgeri [28], Eptatretus stouti [85], and Paramyxine atami [15], and
two species of lamprey: Lampetra japonica [54] and Ichthyomuzon
unicuspis [19]. In addition, some FMRFamide-ir fibers were also
reported in the spinal cord of Lampetra fluviatilis [12]. Some
immunohistochemical studies have provided basic data on the
organization of NPY-like-immunoreactive (NPY-ir) structures
in the brain of cyclostomes, including a hagfish (Paramixine
atami: [14]), and several species of lampreys (Lampetra fluviatilis
and/or Ichthyomyzon unicuspis: [7,11,57,65,79], and Lethenteron
japonica: [17]). In addition, the identity of the peptide in lamprey
has been verified with in situ hybridization [69].
Most immunoreactive cells for these two substances in the
lamprey brain were found in the preoptic and infundibular
hypothalamus. Some small NPY-ir cells were observed in the
subependymal layer of the preoptic area [17]. These cells could
correspond to those NPFF-ir cells found in this research to be
located in the same position. A conspicuous population of
NPY-ir cells was found in the dorsal hypothalamus [17,65].
Most of the FMRF-ir cells were also found in the dorsal
hypothalamus [19,54]. These hypothalamic cells, that were
initially subdivided in three groups (‘‘rostral’’, ‘‘dorsal’’ and
‘‘caudal’’) by Ohtomi et al. [54], and later confirmed by Eisthen
and Northcutt [19], roughly correspond to our anterobasal
nucleus plus bed nucleus of the postoptic commissure. While
this is the only location in the lamprey brain where positive
cells for the three peptides were found; the putative coex-
istence in the same cell should be further explored with double
labeling experiments. As has been shown previously, the cells
initially reported as NPFF-ir in the hypothalamic intermedio-
medial nucleus of rats do not store NPY [35].
In hagfish FMRF-ir cells were almost restricted to hypotha-
lamic structures, including the infundibular hypothalamic
nucleus [15,28,85], and, occasionally, in the surrounding areas.
In addition, NPY-ir cells were also found in the same region in
these animals, and the examination of adjacent sections
immunostained alternatively for NPY and FMRFamide showed
overlapping of the immunoreactive structures, with the same
cells immunostained with both antisera. However, it should be
noted that cross-blocking experiments showed that FMRF-ir
structures could be recognized by the anti-NPY antiserum they
used. In lamprey, there are some other cell populations
immunoreactive only for NPY, such as the dorsal pallium (our
subhippocampal lobe), the preoptic area, the ventral part of
the dorsal thalamus, and the juxtacommissural pretectum
[17,65,90]. Furthermore, in the medial portions of the
mesencephalic tegmentum there are some NPY-ir cells
[11,17] in the same location where NPFF-ir cells were detected.
In the lamprey rhombencephalon, some weakly and
scattered FMRF labeled cells were also seen, in two out of
five animals, in the dorsal isthmal gray [19], which clearly
corresponds to the NPFF-ir dorsal isthmic group we found.
In this study, NPFF-ir cells were also found in the trigeminal
sensory nucleus. FMRF-ir cells were reported in the trigeminal
sensory nucleus of hagfish [85]; however, none were present in
lampreys [19,54]. Concerning the reticular formation, NPY-ir
cells were described in the middle rhombencephalic reticular
nucleus [17]. However, we consider that the NPY-ir cells found
by Chiba [17] in caudal mesencephalic tegmentum (see his
Fig. 2G) to correspond to our anterior rhombencephalic
reticular nucleus (see also [11]). Therefore, NPY-ir cells are
present in both the anterior and the middle rhombencephalic
reticular nuclei, whereas, NPFF-ir cells were only observed in
the caudal part of the anterior rhombencephalic reticular
nucleus. Additional NPY-ir cells were reported in the medial
portions of the rhombencephalic alar plate from the isthmic
region up to the level of the vagal motor nucleus [11,17]. Part of
these cells in the caudal rhombencephalon could correspond
to the nucleus of the solitary tract in the lamprey. Experiments
in the same nucleus of rats demonstrated that neurons
immunoreactive for NPFF and NPY comprise distinct popula-
tions of cells [35].
In the spinal cord of lampreys, FMRFamide-ir cells were not
reported [12,19,54]. However, some NPY-ir cells were observed
p e p t i d e s 2 7 ( 2 0 0 6 ) 1 0 5 4 – 1 0 7 2 1069
in the dorsal part of the cell column [7,11,17,57,79]. In addition,
the NPY-ir fibers appeared to originate from these cell bodies
and were restricted to the dorsal half of the spinal cord [11,79].
Because of NPFF-ir was occasionally seen in the same fibers
with NPY in the porcine spinal cord [83], these substances
could also colocalize to the same neurons. Double labeling
techniques are needed to test this hypothesis.
Fibers immunoreactive for FMRFamide did not form
discrete bundles, but were scattred diffusely throughout
diverse regions of the brain and rostral spinal cord
[12,19,54]. Concerning the neurohypophysis, labeled fibers
were observed in Ichthyomyzon unicuspis [19], but only present
to a limited extend in its anterior part (median eminence) in
Lampetra japonica [54]. In addition, no FMRFamide immunor-
eactivity was found in the neurohypohysis of the three species
of hagfish studied so far [15,28,85]; however, there is a rich
innervation by NPFF-ir fibers. Thus, whereas, the hypotha-
lamic immunoreactive cells for FMRF do not participate in
neuroendocrine mechanisms in hagfishes and their role in
lampreys appears to be very limited, those labeled for NPFF
appear to be highly involved in the regulation of the
neurohypophysis by the hypothalamo-hypophyseal system
(see Section 4.1.2).
4.3. Codistribution of NPFF and catecholamines
In the present study, the presence of NPFF in the catechola-
minergic cells has been investigated, since these substances
are present in neurons that show similar distribution patterns
[59,60,62,84]. If NPFF colocalizes with the catecholamine-
synthesizing enzyme TH, it could act as a cotransmitter in
catecholaminergic neurons and thus be involved in various
somatosensory and visceral functions. Although the NPFF-ir
cells were found in several places where interaction with
catecholaminergic cells could be expected, no colocalization
was found throughout the sea lamprey brain and spinal cord.
However, our findings are of significance in that they add
confirmation to the previously reported subdivisions of the
lamprey preoptic area and hypothalamus [65]. In addition, our
study showed an extensive codistribution of NPFF-ir and TH-ir
fibers in several regions or nuclei in the lamprey CNS. With
this technique, the TH-ir structures (green) and the NPFF-ir
structures (red) are well distinguished in separate neurons.
However, sometimes they appeared somehow masked (show-
ing a yellow-like color), particularly in those areas of high
concentration of labeled fibers such as the neurohypophysis or
the postinfundibular commissure (see Fig. 5A). This is
certainly due to overlapping of fibers with different immunor-
eactivity (TH-ir or NPFF-ir) because of the thickness of the
sections (40 mm), but not because of colocalization of both
substances (TH and NPFF) in the same fiber.
Unfortunately, the presence of double labeled cells for NPFF
and TH in either the isthmic region, the nucleus of the solitary
tract, and the lateral spinal cord could not be studied because
of the absence of immunolabeling with the monoclonal anti-
TH antiserum used in our study. This was somehow expected
because the presence of catecholaminergic cell bodies in these
populations as revealed by using antisera against TH and/or
dopamine has been difficult to demonstrate in lampreys
[59,60,62]. As an example, all the anti-TH antibodies used
previously failed to detect any labeling in the nucleus of the
solitary tract and the lateral spinal cord [59,60,62], whereas,
some labeling was reported in both by using a non-
commercial anti-dopamine antibody [62,68].
5. Conclusions
The comparative analysis of the distribution of FMRF-ir and/
or NPY-ir with that of NPFF-ir structures in the lamprey CNS
showed a partial overlapping which suggests that a NPFF-
like peptide also exists in this animal. Furthermore, the NPFF
immunoreactive network of fibers appears to be well
developed in the brain of the lamprey, a living member of
the most primitive vertebrates. This study reveals that, in
general, the lamprey pattern of NPFF-ir elements is similar to
that previously reported for other vertebrates. In addition, the
organization of some of the NPFF-ir systems in the lamprey
CNS as, for example, the innervation of the neurohypophysis or
the spinal cord, comprises features of the anamniote (amphi-
bian) brain that are also common to all amniotes (mammals)
studied so far, thus being conservative to a surprisingly high
degree. Although more studies are needed, this also suggests
that the function of this neuropeptide could be conserved
through vertebrate evolution. Finally, the double immuno-
fluorescent experiments have shown that, in spite of the high
degree of codistribution found, colocalization of NPFF and TH
was absent in all labeled cell populations.
Acknowledgements
We want to express our gratitude to Dr. H.-Y.T. Yang for his
generous gift of the antiserum used in this study, and Dr. S.
Nagi for copy-editing suggestions. We also thank the Con-
sellerıa de Medio Ambiente of the Xunta de Galicia for their
kind permission to capture of the lampreys used in this study,
as well as M. Garcıa, J. Garcıa and J.A. Garea at the Center for
Control of Fishes of Ximonde (A Coruna). This study was
supported by grants from Ministerio de Ciencia y Technologıa-
FEDER (BFI2003-04479), Xunta de Galicia (PGIDT04P-
XIC31004PN), and University of Vigo (Z441-64102) to MAP,
and Ministerio de Ciencia y Tecnologıa (BFI2003-0559) to AG.
r e f e r e n c e s
[1] Aarnisalo A, Panula P. Neuropeptide FF in the lateral spinaland lateral cervical nuclei: evidence for contacts onspinothalamic neurons. Exp Brain Res 1998;119:159–65.
[2] Aarnisalo A, Tuominen RK, Nieminen M, Vaino P, Panula P.Evidence for prolactin releasing activity of neuropeptide FFin rats. Neuroendocrinol Lett 1997;18:191–6.
[3] Allard M, Theodosis DT, Rousselot P, Lombard MC,Simonnet G. Characterization and localization of a putativemorphine-modulating peptide, FLFQPQRFamide, in the ratspinal cord: biochemical and immunocytochemical studies.Neuroscience 1991;40:81–92.
[4] Auclair F, Lund JP, Dubuc R. Immunohistochemicaldistribution of tachykinins in the CNS of the lampreyPetromyzon marinus. J Comp Neurol 2004;479:328–46.
p e p t i d e s 2 7 ( 2 0 0 6 ) 1 0 5 4 – 1 0 7 21070
[5] Boer HH, Schot LP, Veenstra JA, Reichelt D.Immunocytochemical identification of neural elements inthe central nervous systems of a snail, some insects, a fish,and a mammal with an antiserum to the molluscan cardio-excitatory tetrapeptide FMRF-amide. Cell Tissue Res1980;213:21–7.
[6] Boersma CJ, Sonnemans MA, Van Leeuwen FW.Immunocytochemical localization of neuropeptide FF(FMRFamide-like peptide) in the hypothalamo-neurohypophyseal system of Wistar and Brattlebororats by light and electron microscopy. J Comp Neurol1993;336:555–70.
[7] Bongianni F, Christenson J, Hokfelt T, Grillner S.Neuropeptide Y-immunoreactive spinal neurons makeclose appositions on axons of primary sensory afferents.Brain Res 1990;523:337–41.
[8] Bonini JA, Jones KA, Adham N, Forray C, Artymyshyn R,Durkin MM, et al. Identification and characterization of twoG protein-coupled receptors for neuropeptide FF. J BiolChem 2000;275:39324–31.
[9] Bonnard E, Burlet-Schiltz O, Frances B, Mazarguil H,Monsarrat B, Zajac JM, et al. Identification of neuropeptideFF-related peptides in rodent spinal cord. Peptides2001;22:1085–92.
[10] Bonnard E, Burlet-Schiltz O, Monsarrat B, Girard JP, ZajacJM. Identification of proNeuropeptide FFA peptidesprocessed in neuronal and non-neuronal cells and innervous tissue. Eur J Biochem 2003;270:4187–99.
[11] Brodin L, Rawitch A, Taylor T, Ohta Y, Ring H, Hokfelt T,et al. Multiple forms of pancreatic polypeptide-relatedcompounds in the lamprey CNS: partial characterizationand immunohistochemical localization in the brain stemand spinal cord. J Neurosci 1989;9:3428–42.
[12] Buchanan JT, Brodin L, Hokfelt T, Van Dongen PA, GrillnerS. Survey of neuropeptide-like immunoreactivityin the lamprey spinal cord. Brain Res 1987;408:299–302.
[13] Castro A, Becerra M, Anadon R, Manso MJ. Distribution anddevelopment of FMRFamide-like immunoreactive neuronalsystems in the brain of the brown trout, Salmo trutta fario.J Comp Neurol 2001;440:43–64.
[14] Chiba A, Honma Y, Oka S. Immunohistochemicallocalization of neuropeptide Y-like substance in the brainand hypophysis of the brown hagfish, Paramyxine atami.Cell Tissue Res 1993;271:289–95.
[15] Chiba A, Honma Y. FMRFamide-immunoreactive structuresin the brain of the brown hagfish, Paramyxine atami:relationship with neuropeptide Y-immunoreactivestructures. Histochemistry 1992;98:33–8.
[16] Chiba A. Distribution of neuropeptide Y-likeimmunoreactivity in the brain of the bichir, Polypterussenegalus, with special regard to the terminal nerve. CellTissue Res 1997;289:275–84.
[17] Chiba A. Immunohistochemical distribution ofneuropeptide Y-related substance in the brain andhypophysis of the arctic lamprey, Lethenteron japonica. BrainBehav Evol 1999;53:102–9.
[18] Crespo M, Moreno N, Lopez JM, Gonzalez A. Comparativeanalysis of neuropeptide FF-like immunoreactivity in thebrain of anuran (Rana perezi, Xenopus laevis) and urodele(Pleurodeles waltl) amphibians. J Chem Neuroanat2003;25:53–71.
[19] Eisthen HL, Northcutt RG. Silver lampreys (Ichthyomyzonunicuspis) lack a gonadotropin-releasing hormone- andFMRFamide-immunoreactive terminal nerve. J CompNeurol 1996;370:159–72.
[20] Elshourbagy NA, Ames RS, Fitzgerald LR, Foley JJ, ChambersJK, Szekeres PG, et al. Receptor for the pain modulatoryneuropeptides FF and AF is an orphan G protein-coupledreceptor. J Biol Chem 2000;275:25965–71.
[21] Ferrarese C, Iadarola MJ, Yang HY, Costa E. Peripheral andcentral origin of Phe–Met–Arg–Phe-amide immunoreactivityin rat spinal cord. Regul Pept 1986;13:245–52.
[22] Goossens N, Dierickx K, Vandesande F.Immunocytochemical demonstration of the hypothalamo-hypophysial vasotocinergic system of Lampetra fluviatilis.Cell Tissue Res 1977;177:317–23.
[23] Gouarderes C, Sutak M, Zajac JM, Jhamandas K.Antinociceptive effects of intrathecally administeredF8Famide and FMRFamide in the rat. Eur J Pharmacol1993;237:333–9.
[24] Greenberg MJ, Rao KR, Lehman HK, Price DA, Doble KE.Cross-phyletic bioactivity of arthropod neurohormonesand molluscan ganglion extracts: evidence of an extendedpeptide family. J Exp Zool 1985;233:337–46.
[25] Hinuma S, Shintani Y, Fukusumi S, Iijima N, Matsumoto Y,Hosoya M, et al. New neuropeptides containing carboxy-terminal RFamide and their receptor in mammals. Nat CellBiol 2000;2:703–8.
[26] Jennings JB, Davenport TR, Varndell IM. FMRFamide-likeimmunoreactivity and arylamidase activity in turbellariansand nemerteans-evidence for a novel neurovascularcoordinating system in nemerteans. Comp BiochemPhysiol C 1987;86:425–30.
[27] Jhamandas JH, Jhamandas A, Harris KH. New centralprojections of neuropeptide FF: colateral branchingpathways in the brainstem and hypothalamus in the rat. JChem Neuroanat 2001;21:171–9.
[28] Jirikowski G, Erhart G, Grimmelikhuijzen CJ, Triepel J,Patzner RA. FMRF-amide-like immunoreactivity in brainand pituitary of the hagfish Eptatretus burgeri(Cyclostomata). Cell Tissue Res 1984;237:363–6.
[29] Kavaliers M. Inhibitory influences of mammalianFMRFamide (Phe–Met–Arg–Phe-amide)-related peptides onnociception and morphine- and stress-induced analgesia inmice. Neurosci Lett 1990;115:307–12.
[30] Kavaliers M, Colwell DD. Neuropeptide FF (FLFQPQRF-NH2)and IgG from neuropeptide FF antiserum affect spatiallearning in mice. Neurosci Lett 1993;157:75–8.
[31] Kivipelto L. Ultrastructural localization of neuropeptide FF,a new neuropeptide in the brain and pituitary of rats. RegulPept 1991;34:211–24.
[32] Kivipelto L, Aarnisalo AA, Panula P. Neuropeptide FF iscolocalized with catecholamine synthesizing enzymes inneurons of the nucleus of the solitary tract. Neurosci Lett1992;143:190–4.
[33] Kivipelto L, Majane EA, Yang H-YT, Panula P.Immunohistochemical distribution and partialcharacterization of FLFQPQRFamide-like peptides in thecentral nervous system of rats. J Comp Neurol1989;186:269–87.
[34] Kivipelto L, Panula P. Central neuronal pathwayscontaining FLFQPQRFamide-like (morphine-modulating)peptides in the rat brain. Neuroscience 1991;41:137–48.
[35] Kivipelto L, Panula P. Comparative distribution of neuronscontaining FLFQPQRF-amide-like (morphine-modulating)peptide and related neuropeptides in the rat brain. Eur JNeurosci 1991;3:175–85.
[36] Kivipelto L, Panula P. Origin and distribution ofneuropeptide-FF-like immunoreactivity in the spinal cordof rats. J Comp Neurol 1991;307:107–19.
[37] Kivipelto L, Rubenstein J, Yang HY, Panula P. Ontogeny ofthe F8Famide-like (morphine-modulating) peptides in thecentral nervous system of rats. J Comp Neurol1991;304:14–33.
[38] Kotani M, Mollereau C, Detheux M, Le Poul E, Brezillon S,Vakili J, et al. Functional characterization of a humanreceptor for neuropeptide FF and related peptides.Brit J Pharmacol 2001;133:138–44.
p e p t i d e s 2 7 ( 2 0 0 6 ) 1 0 5 4 – 1 0 7 2 1071
[39] Koyama H, Kishida R, Goris RC, Kusunoki T. Organization ofsensory and motor nuclei of the trigeminal nerve inlampreys. J Comp Neurol 1987;264:437–48.
[40] Kyle AL, Luo BG, Magnus TH, Stell WK. Substance P-,F8Famide-, and A18Famide-like immunoreactivity in thenervus terminalis and retina of the goldfish Carassiusauratus. Cell Tissue Res 1995;280:605–15.
[41] Labrouche S, Verdot L, Theodosis DT, Allard M, SimmonetG. Characterization of a morphine-modulating peptide,FLFQPQRFamide, in the rat hypophysis: biochemical andimmunochemical studies. Endocrinology 1993;132:2191–8.
[42] Lee CH, Wasowicz K, Brown R, Majane EA, Yang HY, PanulaP. Distribution and characterization of neuropeptideFF-like immunoreactivity in the rat nervous systemwith a monoclonal antibody. Eur J Neurosci1993;5:1339–48.
[43] Liu Q, Guan XM, Martin WJ, McDonald TP, Clements MK,Jiang Q, et al. Identification and characterization of novelmammalian neuropeptide FF-like peptides that attenuatemorphine-induced antinociception. J Biol Chem2001;276:36961–9.
[44] Majane EA, Casanova MF, Yang HY. Biochemicalcharacterization of FMRF-NH2-like peptides in spinal cordsof various mammalian species using specificradioimmunoassay. Peptides 1988;9:1137–44.
[45] Majane EA, Panula P, Yang H-YT. Rat brain regionaldistribution and spinal cord neuronal pathway ofFLFQPQRF-NH2, a mammalian FMRF-NH2-like peptide.Brain Res 1989;494:1–12.
[46] Majane EA, Yang HY. Distribution and characterization oftwo putative endogenous opioid antagonist peptides inbovine brain. Peptides 1987;8:657–62.
[47] Majane EA, Yang HY. FMRF-NH2-like peptide is deficient inthe pituitary gland of the Brattleboro rat. Peptides1990;11:345–9.
[48] Majane EA, Yang H-YT. Mammalian FMRF-NH2-likepeptide in rat pituitary: decrease by osmotic stimulus.Peptides 1991;12:1303–8.
[49] Majane EA, Zhu J, Aarnisalo A, Panula P. Origin ofneurohypophyseal neuropeptide FF (FLFQPQRF-NH2).Endocrinology 1993;133:1578–84.
[50] Malin D, Lake JR, Hammond MV, Fowler DE, Brown SL, SimsJL, et al. FMRFamide-like mammalian octapeptide: possiblerole in opiate dependence and abstinence. Peptides1990;11:969–72.
[51] Munz H, Claas B, Stumpf WE, Jennes L. Centrifugalinnervation of the retina by luteinizing hormone releasinghormone (LHRH)-immunoreactive telencephalic neurons inteleostean fishes. Cell Tissue Res 1982;222:313–23.
[52] Nieuwenhuys R, Nicholson C. Lampreys, petromyzontoidea.In: Nieuwenhuys R, ten Donkelaar HJ, Nicholson C, editors.The Central Nervous System of Vertebrates, vol. 1. Berlin:Springer-Verlag; 1998. p. 397–495.
[53] Oehlmann VD, Korte H, Sterner C, Korsching SI. Aneuropeptide FF-related gene is expressed selectively inneurons of the terminal nerve in Danio rerio. Mech Dev2002;117:357–61.
[54] Ohtomi M, Fujii K, Kobayashi H. Distribution ofFMRFamide-like immunoreactivity in the brain andneurohypophysis of the lamprey, Lampetra japonica. CellTissue Res 1989;256:581–4.
[55] Panula P, Aarnisalo AA, Wasowicz K. Neuropeptide FF, amammalian neuropeptide with multiple functions. ProgNeurobiol 1996;48:461–87.
[56] Panula P, Kivipelto L, Nieminen O, Majane EA, Yang H-YT.Neuroanatomy of morphine modulating neuropeptides.Med Biol 1987;65:127–35.
[57] Parker D, Soderberg C, Zotova E, Shupliakov O, Langel U,Bartfai T, et al. Co-localized neuropeptide Y and GABA have
complementary presynaptic effects on sensory synaptictransmission. Eur J Neurosci 1998;10:2856–70.
[58] Perry SJ, Yi-Kung Huang E, Cronk D, Bagust J, Sharma R,Walker RJ, et al. A human gene encoding morphinemodulating peptides related to NPFF and FMRFamide. FEBSLett 1997;409:426–30.
[59] Pierre J, Mabouche M, Suderevskaya EI, Reperant J, Ward R.Immunohistochemical localization of dopamine andits synthetic enzymes in the central nervous system ofthe lamprey Lampetra fluviatilis. J Comp Neurol1997;380:119–35.
[60] Pierre J, Rio JP, Mabouche M, Reperant J. Catecholaminesystems in the brain of cyclostomes, the lamprey, Lampetrafluviatilis. In: Smeets WJA, Reiner A, editors. Phylogeny andDevelopment of Catecholamine Systems in the CNS ofVertebrates. Cambridge: Cambridge University Press; 1994.p. 7–19.
[61] Pinelli C, D’Aniello B, Sordino P, Meyer DL, Fiorentino M,Rastogi RK. Comparative immunocytochemical study ofFMRFamide neuronal system in the brain of Danio rerio andAcipenser ruthenus during development. Brain Res DevBrain Res 2000;119:195–208.
[62] Pombal MA, El Manira A, Grillner S. Afferents of thelamprey striatum with special reference to thedopaminergic system—a combined tracing andimmunohistochemical study. J Comp Neurol1997;386:71–91.
[63] Pombal MA, Marın O, Gonzalez A. Choline acetyltransferaseimmunoreactivity in the hypothalamoneurohypophysialsystem of the lamprey. Eur J Morphol 1999;37:103–6.
[64] Pombal MA, Marın O, Gonzalez A. Distribution of cholineacetyltransferase-immunoreactive structures in thelamprey brain. J Comp Neurol 2001;431:105–26.
[65] Pombal MA, Puelles L. A prosomeric map of the lampreyforebrain based on calretinin immunocytochemistry,nissl stain and ancillary markers. J Comp Neurol1999;414:391–422.
[66] Roth BL, Disimone J, Majane EA, Yang H-YT. Elevations ofarterial pressure in rats by two new vertebrate peptidesFLFQPQRF-NH2 and AGEGLSSPFWSLAAPQRF-NH2, whichare immunoreactive to FMRF-NH2 antiserum.Neuropeptides 1987;10:37–42.
[67] Sasek CA, Elde RP. Distribution of neuropeptide Y-likeimmunoreactivity and its relationship to FMRF-amide-likeimmunoreactivity in the sixth lumbar and first sacralspinal cord segments of the rat. J Neurosci1985;5:1729–39.
[68] Schotland JL, Shupliakov O, Grillner S, Brodin L. Synapticand nonsynaptic monoaminergic neuron systems in thelamprey spinal cord. J Comp Neurol 1996;372:229–44.
[69] Soderberg C, Pieribone VA, Dahlstrand J, Brodin L,Larhammar D. Neuropeptide role of both peptide YY andneuropeptide Y in vertebrates suggested by abundantexpression of their mRNAs in a cyclostome brain.J Neurosci Res 1994;37:633–40.
[70] Sternberger LA. Immunocytochemistry. New York: Wiley;1979.
[71] Stinus L, Allard M, Gold L, Simonnet G. Changes in CNSneuropeptide FF-like material, pain sensitivity, and opiatedependence following chronic morphine treatment.Peptides 1995;16:1235–41.
[72] Sundblom DM, Panula P, Fyhrquist F. Neuropeptide FF-likeimmunoreactivity in human plasma. Peptides1995;16(2):347–50.
[73] Tang J, Yang HY, Costa E. Inhibition of spontaneous andopiate-modified nociception by an endogenousneuropeptide with Phe–Met–Arg–Phe–NH2-likeimmunoreactivity. Proc Natl Acad Sci USA 1984;81:5002–5.
p e p t i d e s 2 7 ( 2 0 0 6 ) 1 0 5 4 – 1 0 7 21072
[74] Tsuneki K. The neurohypophysis of cyclostomes as aprimitive hypothalamic center of vertebrates. Zool Sci1988;5:21–32.
[75] Uchiyama H, Reh TA, Stell WK. Immunocytochemical andmorphological evidence for a retinopetal projection inanuran amphibians. J Comp Neurol 1988;274:48–59.
[76] Ukena K, Iwakoshi E, Minakata H, Tsutsui K. A novel rathypothalamic RFamide-related peptide identified byimmunoaffinity chromatography and mass spectrometry.FEBS Lett 2002;512:255–8.
[77] Vallarino M, Feuilloley M, Thoumas JL, Demorgny R, MasiniMA, Vaudry H. Distribution of FMRFamide-likeimmunoreactivity in the brain of the lungfish Protopterusannectens. Peptides 1995;16:1187–96.
[78] Vallarino M, Salsotto-Cattaneo MT, Feuilloley M, Vaudry H.Distribution of FMRFamide-like immunoreactivity in thebrain of the elasmobranch fish Scyliorhynus canicula.Peptides 1991;12:1321–8.
[79] Van Dongen PA, Hokfelt T, Grillner S, Verhofstad AA,Steinbusch HW, Cuello AC, et al. Immunohistochemicaldemonstration of some putative neurotransmitters inthe lamprey spinal cord and spinal ganglia: 5-hydroxytryptamine-, tachykinin-, and neuropeptide-Y-immunoreactive neurons and fibers. J Comp Neurol1985;234:501–22.
[80] Vesselkin NP, Rio JP, Reperant J, Kenigfest NB, Adanina VO.Retinopetal projections in lampreys. Brain Behav Evol1996;48:277–86.
[81] Vilim FS, Aarnisalo AA, Nieminen ML, Lintunen M,Karlstedt K, Kontinen V, et al. Gene for pain modulatoryneuropeptide NPFF: induction in spinal cord by noxiousstimuli. Mol Pharmacol 1999;55:804–11.
[82] Von Bartheld CS. The terminal nerve and its relation withextrabulbar ‘‘olfactory’’ projections: lessons from lampreysand lungfishes. Microsc Res Tech 2004;65:13–24.
[83] Wasowicz K, Panula P. Distribution of neuropeptide FF inporcine spinal cord in comparison with other neuropeptidesand serotonin. J Comp Neurol 1994;346:530–40.
[84] Weigle C, Northcutt RG. The chemoarchitecture of theforebrain of lampreys: evolutionary implications bycomparisons with gnathostomes. Eur J Morphol1999;37:122–5.
[85] Wicht H, Northcutt RG. FMRFamide-like immunoreactivityin the brain of the Pacific hagfish, Eptatretus stouti(Myxinoidea). Cell Tissue Res 1992;270:443–9.
[86] Wirsig-Wiechmann CR, Basinger SF. FMRFamide-immunoreactive retinopetal fibers in the frog, Rana pipiens:demonstration by lesion and immunocytochemicaltechniques. Brain Res 1988;449:116–34.
[87] Wright DE, Demski LS. Organization of GnRH andFMRFamide systems in two primitive bony fishes (orderpolypteriformes). Brain Behav Evol 1996;47:267–78.
[88] Yang H-YT, Fratta W, Majane EA, Costa E. Isolation,sequencing, synthesis, and pharmacologicalcharacterization of two brain neuropeptides that modulatethe action of morphine. Proc Nat Acad Sci USA1985;82:7757–61.
[89] Yang H-YT, Martin B. Isolation and characterization of aneuropeptide from brain and spinal cord of the rat. SocNeurosci Abstr 1995;21:760.
[90] Yanez J, Pombal MA, Anadon R. Afferent and efferentconnections of the parapineal organ in lampreys: a tracttracing and immunocytochemical study. J Comp Neurol1999;403:171–89.