2 0 X-ray diffraction
-
Upload
sorbonne-fr -
Category
Documents
-
view
1 -
download
0
Transcript of 2 0 X-ray diffraction
X-Ray CrystallographyX-Ray Crystallography The most important technique The most important technique for mineralogyfor mineralogy Direct measurement of atomic Direct measurement of atomic arrangementarrangement
Direct measurement of what was Direct measurement of what was originally deduced from crystal originally deduced from crystal facesfaces
X-RaysX-Rays Electromagnetic radiation – Electromagnetic radiation – similar to visible lightsimilar to visible light = 0.02 to 100 Å= 0.02 to 100 Å = 0.002 to 10 nm= 0.002 to 10 nm Visible light = 400 to 750 nmVisible light = 400 to 750 nm
Fig. 6-6Fig. 6-6
Visible Visible light light spectrumspectrum
Full ra nge of elec tro mag neti c r adia tio nFull ra nge of elec tro mag neti c r adia tio n
(nm) f (hertz)
1 nm = 101 nm = 10-9-9 m m
} X-Rays
X-Ray generationX-Ray generation Heat filament, which discharges electrons
Electron accelerated with 20 to 100 kV toward “target”
Target is Cu or Mo (also Co, Fe and Cr)Very hot – requires continuous circulation of cooling water
Fig. 8-2
Full spectrum of X rays
X-Ray generatedX-Ray generated
Continuous spectrum of Continuous spectrum of X-ray energy X-ray energy (wavelengths) are (wavelengths) are produced without produced without electrons changing electrons changing shellsshells
Characteristic spectrum Characteristic spectrum when incoming electrons when incoming electrons dislodge electrons from dislodge electrons from outer shellsouter shells
Electrons drop from Electrons drop from either M shell (Keither M shell (K) or L ) or L shell (Kshell (K))
KK the most intense the most intense (highest energy)(highest energy)
X-rays
Target material - Cu
Electrons K
K
Fig. 8-3
Use of X-rays requires single wavelengthUse of X-rays requires single wavelength Similar to monochromatic visible lightSimilar to monochromatic visible light Wavelength critical for measurementWavelength critical for measurement Acts like a “ruler”Acts like a “ruler”
Must filter out the continuous spectrum, Must filter out the continuous spectrum, and leave only one of the characteristic and leave only one of the characteristic spectrumspectrum
Typically KTypically K peak – most intense peak – most intense Referred to as Cu – KReferred to as Cu – K radiation radiation
FilteringFiltering Use a monochrometerUse a monochrometer Typically a thin piece of Ni or Be foilTypically a thin piece of Ni or Be foil
Foil allows most Cu-KFoil allows most Cu-K radiation to pass radiation to pass Blocks most wavelengths except KBlocks most wavelengths except K
Absorption Absorption edge, Ni edge, Ni filters out filters out these these wavelengthswavelengths
X-rays pass through filter
X-rays blocked by filter
= 1.5418 Å= 1.5418 Å
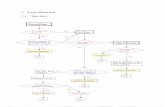
DetectionDetection Variety of detectorsVariety of detectors
Scintillation counters (light Scintillation counters (light flashes)flashes)
Gas proportional countersGas proportional counters Detectors are arranged so that Detectors are arranged so that X-rays reflected off of mineral X-rays reflected off of mineral surfaces can be recordedsurfaces can be recorded
DetectorDetector
SampleSample
Arrangement Arrangement of X-Ray of X-Ray
diffractomediffractometerter
X-ray X-ray generatgenerationion
X-Ray diffractionX-Ray diffraction Wavelength of X rays 1 to 2 ÅWavelength of X rays 1 to 2 Å
Cu KCu K = 1.5418 Å = 1.5418 Å About the same length as About the same length as spacing of atoms in mineralsspacing of atoms in minerals
Typically 1 to 2 ÅTypically 1 to 2 Å
Called d spacingCalled d spacing
= 1.5418 Å
X-Ray diffractionX-Ray diffraction
pqr = npqr = n
pq = d sin pq = d sin
Reflects waves in Reflects waves in phase, only if angle phase, only if angle is such at that the is such at that the additional distance pqr additional distance pqr traveled by wave 2 is traveled by wave 2 is an integer number of an integer number of wavelengths (here 1 wavelengths (here 1 wavelength) wavelength)
Bragg Bragg EquationEquation
pqr = npqr = n
pq = d sin pq = d sin
pqr = 2pq = pqr = 2pq =
2d sin 2d sin = n = n
Bragg Equation
Planes of atoms
ExampleExample Halite – {111} planes have d Halite – {111} planes have d spacing of 3.255 Åspacing of 3.255 Å
Cu KCu K radiation, radiation, = 1.5418 Å = 1.5418 Å Solving Bragg equation showsSolving Bragg equation shows
= 13.70º for n = 1= 13.70º for n = 1 = 28.27º for n = 2= 28.27º for n = 2 = 45.27º for n = 3= 45.27º for n = 3 = 71.30º for n = 4= 71.30º for n = 4
When X-rays reflect off mineral at these angles, they will interfere constructively – cause a peak in energy at the detector
Multiple possible atomic planes {110}, Multiple possible atomic planes {110}, {100}, {001} etc.{100}, {001} etc.
Orienting a single grain unlikely to Orienting a single grain unlikely to reflect X-rays off of any of these reflect X-rays off of any of these planes planes
Better to use multiple grains with Better to use multiple grains with random orientationsrandom orientations With enough planes (1000’s), there will be With enough planes (1000’s), there will be enough parallel to create reflections.enough parallel to create reflections.
Powder Diffraction methodPowder Diffraction method
Powder DiffractionPowder Diffraction Sample crushed to small size, Sample crushed to small size, typically < 0.05 mmtypically < 0.05 mm
Placed on glass slide or hollow Placed on glass slide or hollow holderholder
Sample placed in X-Ray diffractometerSample placed in X-Ray diffractometer Blasted with X-rays as sample and Blasted with X-rays as sample and detector rotate from around 2º to 70ºdetector rotate from around 2º to 70º
Data reductionData reduction
Powder Powder diffraction diffraction filesfiles Cards with the Cards with the intensity and intensity and d spacing for d spacing for all mineralsall minerals
4 major peaks, d spacing
All peaks, d spacings relative intensity, reflecting plane