Population structure of relict Metasequoia glyptostroboides and its habitat fragmentation and...
Transcript of Population structure of relict Metasequoia glyptostroboides and its habitat fragmentation and...
This article appeared in a journal published by Elsevier. The attachedcopy is furnished to the author for internal non-commercial researchand education use, including for instruction at the authors institution
and sharing with colleagues.
Other uses, including reproduction and distribution, or selling orlicensing copies, or posting to personal, institutional or third party
websites are prohibited.
In most cases authors are permitted to post their version of thearticle (e.g. in Word or Tex form) to their personal website orinstitutional repository. Authors requiring further information
regarding Elsevier’s archiving and manuscript policies areencouraged to visit:
http://www.elsevier.com/copyright
Author's personal copy
Population structure of relict Metasequoia glyptostroboides and its habitatfragmentation and degradation in south-central China
Cindy Q. Tang a,⇑, Yongchuan Yang b,c,⇑⇑, Masahiko Ohsawa d, Arata Momohara e, Masatoshi Hara f,Shaolin Cheng g, Shenghou Fan h
a Institute of Ecology and Geobotany, Yunnan University, Kunming 650091, Chinab Faculty of Urban Construction and Environmental Engineering, Chongqing University, Chongqing 400045, Chinac Key Lab of Three Gorges Reservoir Region, Chongqing University, Chongqing 400045, Chinad Institute of Biological Sciences, Faculty of Science, University of Malaya, 50603 Kuala Lumpur, Malaysiae Graduate School of Science, Chiba University, 648 Matsudo, Chiba 271-8510, Japanf Natural History Museum and Institute, Chiba, 9552 Aoba-cho, Chuoku, Chiba 2608682, Japang Hubei Xingdoushan National Nature Reserve Administration Bureau Enshi 445000, Chinah Lichuan Metasequoia Management Office, Lichuan 445400, China
a r t i c l e i n f o
Article history:Received 26 February 2010Received in revised form 1 September 2010Accepted 6 September 2010Available online 29 September 2010
Keywords:Anthropogenic disturbanceConservationMetasequoia glyptostroboidesSize structureSpecies diversity
a b s t r a c t
The relict dawn redwood Metasequoia glyptostroboides Hu et Cheng is endemic to the region borderingHubei and Hunan provinces and Chongqing municipality in south-central China. It is critically endan-gered. We investigated its population size and age structure, and provided a comparison to the studyof Chu and Cooper (1950), documenting the changes of the past 60 years. Our study included all theknown wild individuals of the species as well as analyses of the floristic diversity of their habitats. Inthe last 41 years, habitat changes have effectively ended recruitment of M. glyptostroboides and havereduced species richness in direct response to human disturbance, as shown on several indices. Theremaining specimens ranged from roughly 41 to some 265 years for an average near 95 years, withheights of 12–51 m averaging 27 m. The detrimental activities of human residents include cultivationof profitable plants in the understory, selective cutting, harvesting of wood for fuel, and thoroughgoingcollection of seeds for sale on the active market. Under present environmental conditions and landuse, the dawn redwood will not maintain its natural range in south-central China. Our work detailingthe plant populations in the habitats of this living fossil will be useful in establishing priorities for itsrecovery and conservation.
� 2010 Elsevier Ltd. All rights reserved.
1. Introduction
Human activities across the globe have transformed naturalhabitats into agricultural and artificial landscapes, bringing habitatloss, fragmentation and degradation, and in turn threatening theremaining plant populations. Habitat changes alter the ecology offorest remnants in many ways, reducing the size of species popu-lations and species richness, disrupting their recruitment dynamicsand spatial structure. Habitat degradation cannot only be the directcause of local extinction of plant populations (e.g. Lande, 1988,1998; Tilman et al., 1994; Iida and Nakashixuka, 1995; Clarket al., 1999; Myers et al., 2000). Researchers (e.g. Karron, 1991;Hamrick and Godt, 1996; Tamaki et al., 2008) have alsodemonstrated less immediate effects: endangered species whose
populations are small and isolated are susceptible to loss of with-in-population genetic variation by genetic drift, leading to highextinction rates. The distribution of a species is determined mainlyby its adaptability to the habitat and by barriers to dispersal, aswell as by external events (Cox and Moor, 1993). These consider-ations are borne out in dealing with the topic at hand.
Metasequoia is known as a living fossil, because it was foundonly in fossil records until 1946 (Miki, 1941; Hu, 1946, 1948;Merrill, 1948). According to the fossil history, Metasequoia Mikiwas widely distributed in the Northern hemisphere throughoutthe Tertiary (Florin, 1963; Yu, 1995; Yang and Jin, 2000; Lepageet al., 2005). The relict dawn redwood Metasequoia glyptostroboidesHu et Cheng (formerly Taxodiaceae, and now merged withCupressaceae) currently survives only in wet lower slopes andmontane river and stream valleys in the border region of Hubeiand Hunan provinces and Chongqing municipality in south-centralChina (Cheng and Chu, 1949; Chu and Cooper, 1950; Wang et al.,2005; Leng et al., 2007). Among the IUCN categories of threat,the species is listed as critically endangered (IUCN, 2006). The
0006-3207/$ - see front matter � 2010 Elsevier Ltd. All rights reserved.doi:10.1016/j.biocon.2010.09.003
⇑ Corresponding author. Tel./fax: +86 871 5321702.⇑⇑ Corresponding author. Tel.: +86 138 8331 9188; fax: +86 23 6512 7190.
E-mail addresses: [email protected], [email protected] (C.Q. Tang),[email protected] (Y.C. Yang).
Biological Conservation 144 (2011) 279–289
Contents lists available at ScienceDirect
Biological Conservation
journal homepage: www.elsevier .com/ locate /biocon
Author's personal copy
mountain areas of south-central China harbor flora distinguishedby richness and a high degree of endemism and provide a refugeto many relicts from the Tertiary period, among them Nyssa sinen-sis, Cercidiphyllum japonicum var. sinicum, Tapiscia sinensis andFagus longipetiolata (Chu and Cooper, 1950; Wu, 1980). However,in recent decades the extensive removal of native vegetation inthese areas has left most of the remaining vegetation fragmentedin patches across the landscape. Complex mosaics of stands ofvarying age, composition and structure intermingle with cultivatedfields. An understanding of the structure of M. glyptostroboidespopulations, the species diversity of the communities where thistree is found, and the ecological processes that influence them isimportant for conservation and management. To date there areno studies on the impact of habitat fragmentation and degradationon the population size structure of M. glyptostroboides, althoughmuch excellent research has been conducted from various perspec-tives since the living fossil was discovered in 1940s, including itspaleontology (e.g. Chaney, 1948, 1951; Florin, 1963; Momohara,1994, 2005; Yu, 1995; Yang, 1999; LePage et al., 2005), its contem-porary natural distribution (Hu and Cheng, 1948; Cheng and Chu,1949; Fu and Jin, 1992; Leng et al., 2007), its molecular biologyand genetics (Schlarbaum and Tsuchiyu, 1984; Kuser et al., 1997;Chen et al., 2003; Li, 2006; Yang, 2005), as well as SSRs showingthat the genetic diversity was higher in the wild than in theplanted populations of Metasequoia in China (Li et al., 2005), andthe ecological characteristics (Cheng and Chu, 1949; Chu and Coo-per, 1950; Gressitt, 1953; Li and Ban, 1989; Williams et al., 2003;Yang et al., 2004; Williams, 2005; You et al., 2008).
Taking into account that the size structure of populations pro-vides an indication of their regeneration process, we analyzed thepopulation structure in the fragmented landscape using data fromfield investigation done during 2007–2009. Understanding speciesdiversity of the mosaic M. glyptostroboides communities as well ashabitat fragmentation is important for evaluation of the local threa-tened status because species in a community often interact and de-pend on each other. The present work was performed to determinethe size structure of M. glyptostroboides populations in relation tothe species’ natural status, as well as the impact of habitat loss, frag-mentation and degradation on the species diversity of its communi-ties. Our objectives were: (1) to analyze the structure of wild M.glyptostroboides populations to determine whether recruitmentsoccurred in the various habitats, (2) to evaluate how stand dynamicshave changed in the past 60 years by comparison with the results ofChu and Cooper (1950), and (3) to recommend management andconservation efforts.
2. Methods
2.1. Study areas
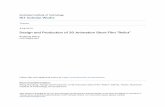
All areas were investigated where wild M. glyptostroboides hadpreviously been recorded (Leng et al., 2007), including Lichuancounty of Hubei province, Shizhu county of Chongqing municipal-ity, and Longhshan county of Hunan province, in order to detectextant populations. The three areas are located in the Yangtze Rivervalley (Fig. 1a). The altitudinal distribution range of wild M. glypto-stroboides populations is from 800 to 1510 m above sea level. On alarge geographic scale, this altitudinal range belongs both to thesubtropical moist evergreen broad-leaved forest zone and the ever-green, deciduous broad-leaved mixed forest zone, with a humid,warm climate. Lichuan, Hubei is the distribution center of the wildliving tree, while only a few individuals are found in Shizhu, Chon-gqing, and in Longshan, Hunan. The three areas have similar histo-ries of agricultural development and forest management, accordingto unpublished information from the local forestry office. All the
habitats in these areas have been disturbed, primarily by increas-ing numbers of village houses, rice paddies, cornfields, and the cul-tivation of Coptis chinensis, a cash medicinal herb that is commonlygrown in the understory of natural or secondary forests. In Lichuan,about 90% of the wild M. glyptostroboides are found in the Zhongluarea, c. 60 km southwest of the Yangtze River. Zhonglu is located atthe confluence of the Xiaohe river valley (the Shuisha valley) andthe Shiziba river valley (Fig. 1b). The valleys are bounded on thewest by Chiyaoshan, a range of Permian limestone which reachesan altitude of 1500 m, and the rivers’ headwaters are largely in thatrange. On the east is a roughly parallel range of Jurassic sandstone,Fubaoshan, which attains an altitude of 1400 m (Chu and Cooper,1950). Many of the wild M. glyptostroboides are located in frag-mented forests near streams. The soil depth is from 35 to 80 cmin the fragmented and disturbed forest habitat. The pH valuesrange between 6.01 and 6.19 for the surface soil (0–15 cm).According to our field soil samples from the study sites, organicmatter ranges from 2.9% to 7.4% of the surface soil. Outcrops arecommon and the forest floor is very moist. The micro-topographyis typified by Gangchangwan, located in Xiaohe (Fig. 1c).
The modern climate of the three areas, as typified by Lichuan(Fig. 2) is humid subtropical, and is largely controlled by the EastAsian monsoon. According to data taken from 1958 to 2000(MBL, 1958–2000), the mean annual temperature is 12.7 �C. Duringthe summer, the region is occupied by a subtropical high pressuresystem and experiences warm, wet conditions, with a highestmonthly mean temperature of 22.6 �C in August; during the win-ter, on the other hand, the area is under the influence of the Mon-golian high pressure system and experiences relatively coldconditions (sometimes with snow) with an average minimum tem-perature of 1.9 �C in January. The monthly relative humidity ishigh, fluctuating between 81% and 83%. The mean annual evapo-transpiration rate is 1071.9 mm. Mean annual precipitation is1319 mm, of which about 85% falls between April and October.
2.2. Data collection and analysis
Between 2007 and 2009, we marked all the wild trees (in total5371 individuals) of M. glyptostroboides with numbered tags, andmapped their location. We obtained 86 increment cores from wildtrees of different age classes. Tree ages were determined using thesoftware WinDENRO tree ring analysis system (Canada). Because ofthe buttress at the base of the trunk, the cores were taken fromabove ground level at 1.3 m, and the length of time missed toground level was estimated as 5 years, corresponding to the heightof the seedlings in a nearby nursery.
In Zhonglu, we randomly selected 31 plots in the patches con-taining M. glyptostroboides at altitudes ranging from 900 to1400 m. The plot areas varied from 600 to 2000 m2 depending onthe size of the patches and the maximum number of species. Weused the abundance of relative basal area method (Ohsawa,1984; Kikvidze and Ohsawa, 2002) for our vegetation sampling.In studying the overstory, a species inventory was carried out forall the individuals at least 1.3 m high in each plot. All were taggedwith number tape, recorded with species name, whether healthy,unhealthy or dead, diameter at breast height (DBH) and tree height(H). The tree stems were classified into the following three groupsbased on the vertical position, crown position and height: the can-opy layer, P18 m tall; the subcanopy layer, below the canopy layerand P8 m tall; and the shrub layer between 1.3 m and 8 m tall. Forthe understory, all woody species (5 cm 6 H < 50 cm for seedlings,50 cm 6 H < 130 cm for saplings) occurring within each plot wereidentified, measured by height and coverage, and counted. Herbspecies were identified, and the coverage and the abundance ofeach herb species were recorded for each plot. Relative Sherensen’ssimilarity index and group average clustering of the plots were
280 C.Q. Tang et al. / Biological Conservation 144 (2011) 279–289
Author's personal copy
used for similarity dendrogram analysis. The values of relative ba-sal areas for each species greater than 1.3 m tall were applied inthe similarity dendrogram analysis.
The diversity indices for woody species were calculated usingFisher’s alpha (Williams, 1947), the Shannon–Wiener index (H0)(Pielou, 1969) and the reciprocal of the Berger–Parker index (1/d)(Magurran, 1988). Fisher’s alpha is a satisfactory scale-indepen-dent indicator of biodiversity. However, alpha may be underesti-mated in communities in which the spatial arrangement ofindividuals is strongly clustered or in which the total number ofspecies does not tend to infinity. When a maximum number of spe-cies is sampled in a habitat, the Shannon–Wiener index provides a
good learning tool for comparing distinct habitats. The Berger–Parker index is simply the number of individuals in the dominantspecies of relation, which allows us to consider the most dominantspecies in their different habitats. Differences in species richnessand diversity among habitats were analyzed by the non-paramet-ric Kruskal–Wallis all-pairwise comparisons test using Analyze-itsoftware (United Kingdom).
Based on the number of human disturbance factors (planting ofprofitable species in the understory, selective cutting, harvesting ofwood for fuel, seed collection, creating agriculture lands such asrice paddies, corn fields, and building roads and houses), each sitewas assigned one of the following disturbance values: 3, if there
Yangtze RiverWanzhou
Shizhu
Longshan
LichuanEnshi
Luota
TaniSangzhi
Shazi
Huangshui
Jiannan
Zhonglu
FubaoshanLiangwu
Wangying
Maodao
29oN
30oN
31oN110oE108oE 109oE
Zhonglu
Yu R
iver
Xiaohe Shizhiba
Qiy
aosh
an(M
t. Q
iyao
)
Fubaoshan
02km
(Mt. Fubao)
Karst upland
Fragmented and disturbedhabitats of M. glyptostroboides
Xiaohe River
Shiziba RiverGuihua
2m
4m
6m
8m
10m
12m
0mStream in themountain slope
M. glyptostroboides
5m
(a) (b)
(c)
Fig. 1. The distribution areas in Shizu, Lichuan and Longshan, south-central China (a). Zhonglu town (b). The representative micro-topography (contours at 1 m intervals) of amountains slope in Gangchangwan of Xiaohe, Zhonglu (c).
C.Q. Tang et al. / Biological Conservation 144 (2011) 279–289 281
Author's personal copy
was only one disturbance factor; 5, if there were two factors; 11, ifthere were three factors; 15, if there were four factors and 30, ifthere were more than four factors. We classified habitats basedon their disturbance scores: habitats with scores of 5–15 werecalled Habitat A (HA) and those with scores of 30 were called Hab-itat B (HB). We further divided the fragmented and disturbed hab-itat (HA) into three categories: Habitat A1 (with the score of 5 asthe degree of disturbance), Habitat A2 (with a score of 11 as the de-gree of disturbance), Habitat A3 (with a score of 15 as the degree ofdisturbance). The representative habitats are shown in photos #1–5 as electronic Supplemental data.
3. Results
3.1. M. glyptostroboides trees and their habitats
All the wild trees of M. glyptostroboides are known. The censusobtained for the three areas is shown in Table 1. For Lichuan, Hu-bei: 5371 plants excluding unhealthy ones, among them the firstspecimen found in Maodaoqi in 1947, of 248 cm DBH, excludedas unhealthy. Trees (2107) were distributed in fragmented and dis-turbed forest in valley bottoms and by streams (HA with scores of5–15 as the degree of human disturbance). Trees (3231) were scat-tered along rice paddies, roadsides, or yards of village houses (HBwith scores of 30 as the degree of human disturbance). In Shizhu,
Chongqing: 28 plants were found in this location, scattered alongrice paddies or roads or yards of village houses (HB). In Longshan,Hunan: five individuals were found, by rice paddies or roads oryards of village houses (HB). No natural seedlings or saplings werediscovered in any of the habitats.
3.2. Size structure
The diameters at a height of 1.3 m (DBH) and correspondingages of wild M. glyptostroboides in Zhonglu, Lichuan are shown inFig. 3. The relationships (y = 1.4x + 6.03, r2 = 0.69, n = 86) indicatea correlation between DBH and age. The growth rate was highlyvariable, and according to ring width for the 86 samples, generallydecreased with advancing age. When the trees were under80 years old, the average growth rate (average ring width) declinedfrom 7.81 mm to 2.13 mm per year. For trees 80–159 years old therate was 2.42 mm to 0.96 mm per year. When the trees were be-tween 160 and 265 years old the rate was 1.89 mm–0.34 mm peryear. The average tree ring data among the 86 sample trees withstandard deviation are shown in Appendix 1.
The DBH data for M. glyptostroboides populations revealed theabsence of young individuals (Fig. 4a–d). In the fragmented anddisturbed forests of Lichuan, no individuals less than 25 cm DBHwith an age of c. 41 years were found (Fig. 4a). In the habitats byrice paddies or near roadsides or by the yards of village houses,there were no trees with a DBH less than 35 cm and none less than55 years old (Fig. 4b). Maximum DBH recorded was 165 cm, whichcorresponds to an age of 237–265 years. Most trees were in theDBH-classes of 45–90 cm, around 69–132 years old. A few individ-uals greater than 40 cm DBH and c. 62 years old were found alongrice paddies or roadsides or in yards of village houses in Shizhu(28) or Longshan (5) (Fig. 4c–d). The height of all the trees rangedfrom 12 to 51 m with an average of 27 m.
The variations of number of individuals in DBH-classes amongplots of Habitat A are shown in Table 2. There were more M. glypto-stroboides trees in 25–45 cm DBH in HA1 than in HA2 and HA3,though most individuals of the species in the three habitats wereof 45–85 cm DBH, around 69–125 years old.
3.3. Communities’ characteristics
For our 31 plots in Zhonglu of Lichuan, the dendrogram showsthree groups at 55% of floristic similarity (Fig. 5a). Habitat A1(HA1 including 13 plots) was relatively natural, with a score of 5as the degree of disturbance, and the subcanopy was dominatedby evergreen and deciduous broad-leaved trees including Phoebe,Cyclobalanopsis, Machilus, Ficus, Liquidambar, Cornus, Castanea andconiferous Cunninghamia, as well as cultivated coniferous Crypto-
J F J AM A M J S O N D0
20
40
60
80
100
300
0
10
20
30
40
50
Tem
pera
ture
(o C
)
Prec
ipita
tion
(mm
)
MAT 12.7 oC
MAP 1318.7 mm
Month
Altitude 1075 m
Lichuan
30°18'15" N108°57'52" E
Fig. 2. Eco-climatic diagram of Lichuan. MAT: mean annual temperature; MAP:mean annual precipitation. Data source is Meteorological Bureau of Lichuan1958–2000.
Table 1Number of individuals of wild M. glyptostroboides in the three areas of south-centralChina. We classified the Habitat according to the degree of disturbance: Habitat A(HA) with scores of 5–15. Habitat B (HB) with a score of 30.
Location Habitat type No. ofadultplantsa
No. ofsaplings/seedlings
Lichuan of Hubei HA: fragmented and disturbed forestsnear streams
2107 0
HB: by rice paddies, corn fields,roadsides or yards of village houses
3231 0
Shizhu ofChongqing
HB: by rice paddies, corn fields,roadsides or yards of village houses
28 0
Longshan ofHunan
HB: by rice paddies, corn fields,roadsides or yards of village houses
5 0
Total 5371 0
a The number of adult plants only includes healthy individuals of wild M.glyptostroboides.
20 60 100 140 18040
80
120
160
200
240
280
Age
(ye
ar)
DBH (cm)
y = 1.4x + 6.03
r2 = 0.69n = 86
Fig. 3. Relationships between diameter at a height of 1.3 m (DBH) and age at thebase of the trunk for wild M. glyptostroboides in Zhonglu, Lichuan.
282 C.Q. Tang et al. / Biological Conservation 144 (2011) 279–289
Author's personal copy
meria, while the shrub and understory were composed of Camellia,Hydrangea, Eurya, Spiraea, etc. Habitat A2 (HA2 including six plots)had a score of 11 as the degree of disturbance; Cryptomeria hasbeen planted since the 1960s, and the shrub layer and understorywere dominantly Camellia, Berberis, Rhus and Rubus. Habitat A3(HA3 including 12 plots) had a score of 15 as the degree of distur-bance, and bamboos were frequently cultivated, including Neosin-ocalamus affinis, Phyllostachys heteroclada and Phyllostachyssulphurea, with some medicinal herbs C. chinensis in the under-story. In habitat A1 (HA1), the stands were multilayered, contain-ing canopy, subcanopy, shrub and understory layers (Fig. 5b). Inhabitat A2 (HA2), the shrub layer was sparse. In habitat A3(HA3), subcanopy and shrub layers were absent. The coverage ofherbaceous species in HA1 was 70–100%, including Impatiensdicentra, Elatostema involucratum, Galium odoratum, Viola vaginata,Polygonum hydropiper, Iris tectorum, etc. and various ferns such asParathelypteris japonica, Selaginella tamariscina, Polystichum neolob-atum, Coniogramme intermedia. The herbaceous coverage in HA2and HA3 was only between 10% and 30%. All the M. glyptostroboideswere over 16 m tall in the three habitats, except for one just 13 mtall in HA1. No seedlings or saplings of M. glyptostroboides existedin these habitats.
3.4. Recruitment
To compare with the data taken in 1948, we adapted the cate-gories of Chu and Cooper (1950) in Fig. 6. Class I was for the indi-
viduals with a height less than 30.5 cm. Class II was with a heightgreater than 30.5 cm but a diameter at breast height (DBH) lessthan 2.5 cm. Class III was 2.5–7.6 cm DBH. Class IV was 10.2–25.4 cm DBH. Class V was greater than 25.4 cm DBH. Individualsof M. glyptostroboides were only found greater than 25 cm DBH(Class V) in the fragmented and disturbed habitats (HA) in 2009(Fig. 6a), whereas individuals had been distributed in classes rang-ing from class I to class V in the natural habitat in 1948 (Fig. 6b).
3.5. Species diversity in communities where M. glyptostroboides wasfound
In the various mosaic plant communities containing M. glyptost-oboides, 91 woody species in total were recorded. Cultivated plantsincluding Cryptomeria fortunei were common in the present habi-tats (Table 3). Habitat A1, with a score of five indicating a low de-gree of human interference, had significantly higher values ofspecies richness and diversity as seen in the reciprocal of the Ber-ger–Park index, Shannon’s H’ and Fisher’s alpha, than habitats A2(HA2) and A3 (HA3) with scores of 11 as moderate and15 as highdegrees of human disturbance (Fig. 7a–d), though each habitathad rather low diversity indices. Species richness was 18–27 perstand for HA1, 6–14 per stand for HA2 and 2–8 per stand forHA3. The reciprocal of the Berger–Park index significantly de-creased from 1.288 in HA1 to 1.161 in HA2, then to 1.018 inHA3. Shannon’s H’ significantly varied between 1.123 and 0.113among the habitats. Fisher’s alpha had the significantly higher va-
0
2
4
0 10 20 30 40 50 60 70 80 90 100
110
120
130
140
150
160
170
HB
Num
ber
of in
divi
dual
s
DBH-class (cm)
(c) Shizhu of Chongqing
DBH-class (cm)
0 10 20 30 40 50 60 70 80 90 100
110
120
130
140
150
160
170
(d) Longshan of Hunan
HB
HA
20
40
60
80
100
120
140
160
180
200
220
240
260
280
300
320
340
360
380
400
420
440
0
Num
ber
of in
divi
dual
s
Lichuan of Hubei(a) (b) Lichuan of Hubei
HB
Fig. 4. The frequency of distribution in DBH-class for M. glyptostroboides in Lichuan (a and b), Shizhu (c) and Longshan (d). HA: fragmented and disturbed forests nearstreams; HB: by rice paddies or corn fields or roadsides or yards of village houses.
C.Q. Tang et al. / Biological Conservation 144 (2011) 279–289 283
Author's personal copy
Tabl
e2
Out
line
ofth
est
udy
plot
san
dnu
mbe
rof
indi
vidu
als
wit
hdi
ffer
ent
DBH
-cla
sses
for
M.g
lypt
ostr
oboi
des
inea
chpl
otin
Zhon
glu,
Lich
uan.
Hab
itat
Plot
Plot
area
(m2)
Loca
lsi
ten
ame
N.L
at.
E.Lo
ng.
Ref
eren
cen
o.a
DB
H-c
lass
(cm
)
25–3
4.9
35–4
4.9
45–5
4.9
55–6
4.9
65–7
4.9
75–8
4.9
85–9
4.9
95–1
04.9
HA
1P
190
0M
aoba
zi30
�100
900
108�
360 4
7.100
CLC
2302
664
22
33
72
P2
450
Gan
gch
angw
an30
�100
7.300
108�
360 4
0.200
CLC
1301
578-
864
2P
340
0G
enpa
nw
an30
�100
3.700
108�
360 3
7.300
CLC
1302
519-
312
52
1P
2790
0H
uos
han
gou
30�0
9059
.800
108�
360 0
10C
LC13
0085
6-62
13
11
1P
1260
0Fe
nsh
anw
an30
�80 1
7.500
108�
360 2
.300
CLC
2302
643
21
12
P15
300
Wu
jipi
ng30
�100
20.200
108�
360 4
900
CLC
1300
842-
72
12
21
P17
400
Ban
zhu
lin
30�80 4
2.400
108�
360 1
9.700
CLC
2302
650
14
P14
1800
Daw
an30
�90 1
3.400
108�
370 1
2.300
CLC
2302
659
54
6P
1860
0X
ingy
un
zili
angs
han
30�80 2
3.100
108�
360 1
3.800
CLC
2302
645
11
32
1P
2012
00Sh
ash
uw
an30
�80 2
1.300
108�
360 1
1.100
CLC
2302
644
15
22
P19
600
Xin
gtia
nw
an30
�80 4
0.700
108�
360 5
4.500
NA
23
11
P6
400
Shan
gsh
ich
angg
ou30
�80 5
1.300
108�
360 1
8.700
CLC
2302
651
12
2P
2180
0Q
ish
upi
ng
30�70 3
5.300
108�
360 1
7.200
CLC
2302
611
14
23
11
HA
2P
1190
0Fe
npi
ng
30�80 4
4.300
108�
360 1
2.500
NA
13
21
P16
300
Das
hit
ouw
an30
�90 3
3.9300
108�
360 3
6.500
NA
21
1P
2380
0Ta
osh
uw
an30
�700
500
108�
360 8
.600
NA
23
13
11
1P
3080
0Y
iaoz
iwan
30�1
0018
.500
108�
360 4
6.700
NA
55
41
11
P24
200
Mao
bazi
shu
ijin
glia
ngs
han
30�1
0011
.400
108�
360 4
1.200
NA
11
1P
2940
0B
eife
npi
ng
30�80 3
1.700
108�
360 2
.400
NA
21
HA
3P
830
0Y
ouji
awan
30�0
5048
.100
108�
370 2
2.600
CLC
2302
704
6P
3180
0La
ofen
zui
30�60 5
4.600
108�
360 5
.100
NA
35
12
1P
2230
0Y
angj
iah
eheb
ian
30�1
0025
.100
108�
410 3
5.800
CLC
2302
610
21
2P
980
0G
uh
ongs
han
wu
qian
heb
ian
30�1
0020
.600
108�
390 4
000
CLC
2302
589
11
41
11
P10
600
Shu
ish
ayu
anw
an30
�100
26.800
108�
390 5
8.100
CLC
2302
588
21
2P
2640
0G
uih
axia
oxu
e30
�080
55.600
108�
360 2
9.600
CLC
2302
652
21
11
P5
400
Shan
gmao
hes
han
30�0
7017
.200
108�
390 1
.600
NA
11
1P
480
0B
aoji
awan
30�0
6043
.300
108�
370 5
.100
CLC
2302
623
34
31
P25
600
Shu
ijin
gwan
30�0
901.
100
108�
360 4
7.300
CLC
1300
875
33
41
1P
760
0X
iash
ich
angg
ou30
�080
45.900
108�
360 2
1.300
CLC
2302
651
12
12
1P
2880
0M
aobi
lian
ggou
30�0
909.
600
108�
370 0
500
CLC
2302
658
14
31
P13
2000
Fen
wan
30�0
9015
.400
108�
360 1
7.800
CLC
1301
528-
303
64
2
NA
:n
otav
aila
ble,
not
fou
nd
inth
eci
ted
sou
rce.
aIt
refe
rsth
ere
fere
nce
nu
mbe
rof
Len
get
al.,
2007
.
284 C.Q. Tang et al. / Biological Conservation 144 (2011) 279–289
Author's personal copy
lue of 18.419 in HA1 as compared with those in HA2 (5.087) andHA3 (1.703). The statistical test was checked by the non-paramet-ric Kruskal–Wallis all-pairwise comparisons and the significancevalues were at p < 0.001.
4. Discussion
4.1. Recruitment
Land clearance and conversion to agriculture and plantation for-estry have led to loss of actual and potential habitats in these areas.The existing habitats of M. glyptostroboides have been exposed to dis-
turbance, principally by cultivation of other plants and by seed col-lecting. Seedlings are often at an especially sensitive stage in theplant life cycle, requiring considerably more specific conditions thanadults to support their growth. In many plant species, the largest lossof reproductive potential occurs between seed and seedling estab-lishment. Biotic and abiotic factors acting during early recruitmentmay have profound effects on the dynamics and spatial structureof a population (Cavers and Harper, 1967; Schupp and Fuentes,1995; Clark et al., 1999). Seedlings and saplings of M. glyptostroboideswere found in the dense and shady understory in the ecological sur-vey of 1948 (Chu and Cooper, 1950). But neither seedlings nor sap-lings were discovered in the current environment. This speciesshows no ability to recover from human disturbance in these areas.
Plot
7
Plot
13
Plot
25
Plot
28
Plot
22
Plot
4
Plo
t 8
Plot
9
Plot
31
Plot
5Pl
ot 2
6Pl
ot 1
0
Sim
ilari
ty (
%)
Canopy
Subcanopy
& understory
Intensity ofdisturbance HighModerateLow
Habitat
Metasequoia
Metasequoia Metasequoia
CryptomeriaLiquidambarCornusPhoebe
FicusCastanea
CryptomeriaCunninghamia
Eurya BerberisDaphne
HydrangeaSpiraeaStachyurus
Decaisnea RubusRhus
PhyllostachysCamellia
Cyclobalanopsis
Neosinocalamus
Machilus
HA3HA2HA1
Plo
t 1Pl
ot 2
Plot
3
Plo
t 6
Plot
11
Plot
12
Plot
14
Plot
15
Plot
16
Plot
17
Plot
18
Plot
19
Plot
20
Plot
21
Plot
23
Plot
24
Plot
27
Plot
29
Plot
30
Rel
ativ
e no
. of
indi
vidu
als
(%)
Height-class (m) Height-class (m) Height-class (m)0 8 16 24 32 40 0 8 16 24 32 40
Metasequoia
(a)
(b)
0 8 16 24 32 400
10
20
30
40
50
HA1 HA2 HA3
Shrub layer
Trachycarpus
0
25
50
75
100
LiquidambarCornus
Rhus
Camellia
Rubus
Fig. 5. Similarity dendrogram using relative Sherensen’s similarity index and group average clustering (a), and the frequency of distribution in height-class for all the plantsover 1.3 tall in habitat A1 (HA1), habitat A2 (HA2) and habitat A3 (HA3) (b). Black bar indicates M. glyptostroboides, blanked bar is for cultivated Cryptomeria fortunei, grey baris for the other species.
C.Q. Tang et al. / Biological Conservation 144 (2011) 279–289 285
Author's personal copy
Table 3Floristic composition (woody species, bamboo and Trachycarpus with RBA P 0.08%) in the fragmented and disturbed stands in Zhonglu, Lichuan. the species are ordered bydecreasing value of relative basal area, except the species in HA2 and HA3 which appeared in more than one habitat.
Habitat HA1 HA2 HA3Score of degree of disturbance 5 11 15Altitude (m) 1000–1300 1000–1301 900–1330Number of plots 13 6 12Area of plots (m2) 9350 3400 8400Relative basal area (RBA) RBA (%) RBA (%) RBA (%)
Dominant speciesMetasequoia glyptostroboides Hu et Cheng 77.9 86.1 98.3
Companion speciesLiquidambar formosana Hance in Ann. 4.9 0.9 0.1Cornus controversa Hemsl. 4.3 0.4 0.1Machilus pingii Cheng ex Yang 2.3Phoebe hunanensis Hand.-Mazz. 2.1Cryptomeria fortunei Hooibrenk. ex Otto et Dietr. 1.7 6 0.4Cunninghamia lanceolata Hook. 1.3 5.2 0.3Ficus heteromorpha Hemsl. 0.9Cyclobalanopsis multinervis Cheng et T. Hong 0.7Euscaphis japonica (Thunb.) Kanitz 0.7Castanea seguinii Dode 0.6Diospyros sp. 0.5Lindera pulcherrima (Wall.) Benth. var. attenuata Allen 0.4Betula sp. 0.3 +Acer sinense Pax 0.3Prunus padus L. 0.2Corylus sp. 0.1Rhus potaninii Maxim. 0.1 +Vernicia fordii (Hemsl.) Airy Shaw 0.1 +Toxicodendron succedaneum (L.) O. Kuntze var. succedaneum + 0.8Broussonetia papyrifera (L.) L’Herit. ex Vent. + +Xylosma racemosum (Lour.) Merr. +Styrax japonicus Sieb. et Zucc. +Eurya hebeclados Ling +Hydrangea villosa Rehd. +Quercus serrata Thunb. var. brevipetiolata (A. DC.) Nakai +Acer davidii Franch. +Cinnamomum wilsonii Gamble +Camellia oleifera Abel. +Camellia caudata Wall. +Abelia chinensis R. Br. +Lindera glauca (Sieb. et Zucc.) Bl. +Camellia japonica L. +Viburnum erosum Thunb. +Lindera aggregata (Sims) Kosterm. +Dendrobenthamia angustata (Chun) Fang +
Companion speciesTaxus chinensis (Pilger) Rehd. var. mairei (Lemee et Levl.) +Cheng et L.K. FuMagnolia officinalis Rehd. et Wils. +
1948
I II III IV V
(b)
0102030405060708090
100
Rel
ativ
e nu
mbe
r of
indi
vidu
als
(
%)
I II III IV V
2009
(a)
Fig. 6. The frequency of distribution in DBH-class for M. glyptostroboides in the fragmented and disturbed habitat (HA) in 2009 (a) and in the natural habitat in 1948 (b). ClassI: height less than 30.5 cm; Class II: height greater than 30.5 cm but diameters at breast height (DBH) less than 2.5 cm; Class III: 2.5–7.6 cm DBH; IV: 10.2–25.4 cm DBH; V:DBH greater than 25.4 cm. Data for 1948 are from ten 100 m2 plots of Chu and Cooper (1950).
286 C.Q. Tang et al. / Biological Conservation 144 (2011) 279–289
Author's personal copy
4.2. Species diversity in communities where M. glyptostroboides wasfound
In 1948 the floristic composition in the natural habitat of M. gly-ptostroboides included a number of relicts of the Tertiary period,
among them N. sinensis, C. japonicum var. sinicum, T. sinensis andF. longipetiolata (Chu and Cooper, 1950). Those species were absentat the present time (Table 3). On the other hand, profitable culti-vated plants are commons including C. fortunei, N. affinis, P. heteroc-lada, P. sulphurea, Trachycarpus fortunei and C. chinensis. The species
Table 3 (continued)
Habitat HA1 HA2 HA3Score of degree of disturbance 5 11 15Altitude (m) 1000–1300 1000–1301 900–1330Number of plots 13 6 12Area of plots (m2) 9350 3400 8400Relative basal area (RBA) RBA (%) RBA (%) RBA (%)
Decaisnea insignis (Griff.) Hook. f. et Thoms. +Dichroa febrifuga Lour. + +Cephalotaxus fortunei Hook. +Rhus chinensis Mill. + +Stachyurus chinensis Franch. +Toricellia angulata Oliv. + +Morus australis Poir. +Pittosporum glabratum Lindl. var. neriifolium Rehd. +Euonymus alatus (Thunb.) Sieb. +Daphne odora Thunb. +Hydrangea aspera D. Don + +Trachycarpus fortunei (Hook.) H. Wendl. + + +Dalbergia dyeriana Prain ex Harms in Engl. +Robinia pseudoacacia Linn. +Rhamnus utilis Decne. +Quercus sp. +Idesia polycarpa Maxim. +Glochidion wilsonii Hutch. + +Rhododendron sp. +Cotoneaster divaricatus Rehd. +Spiraea japonica L.f. var. fortunei (Planchon) Rehd. +Coriaria nepalensis Wall. + +Berberis bergmanniae Schneid. + +Debregeasia orientalis C.J. Chen + +Phellodendron chinense Schneid. + +Nothopanax davidii Harm. +Desmodium racemosum (Thunb.)DC. +Rubus flagelliflorus Focke ex Diels + + +Phyllostachys bambusoides Sieb. et Zucc. + +Phyllostachys heteroclada Oliver. + +Phyllostachys sulphurea (Carr.) A. et C. Riv. +Neosinocalamus affinis (Rendle) Keng f. +Bambusa surrecta Q.H. Dai + +
p < 0.0001
Spec
ies
rich
ness p < 0.0001
0.95
1.05
1.15
1.25
1.35
p < 0.0011.61.41.2
10.80.60.40.2
0-0.2
0
510
1520
25
30
p < 0.05
a
b b05
101520253035
HA1 HA2 HA3 HA1 HA2 HA3
Habitat Habitat
a
a
a
b
b
b
c c
c
Shan
non-
Wie
ner
H’
Fish
er’s
alp
haR
ecip
roca
l of
Ber
ger-
Park
inde
x
(a) (b)
(c) (d)
Fig. 7. Species richness (a) and diversity indices in the fragmented and disturbed habitats (c–d). Significantly different values are indicated by different letters, p < 0.00001.
C.Q. Tang et al. / Biological Conservation 144 (2011) 279–289 287
Author's personal copy
diversity of the plant communities containing M. glyptostroboideshas declined along with increasing human encroachment. In Hab-itat A3 (HA3), the associated plant species were almost completelyremoved during the cultivation of bamboos and some medicinalherbs in the understory. This may explain why the diversity is verylow in HA3 (Fig. 7).
Chu and Cooper (1950) recounted that according to local tradi-tion (through interviews with elderly people) 200 years ago themountain slope and the valley bottom were covered by forests.When the earliest permanent settlers migrated to the Shuisha(Metasequoia) valley, they used fire to destroy the forests andestablished rice paddies. Rice culture since then has spread overthe whole level valley floor, and the forests on the mountain slopeshave been largely destroyed to make timber and charcoal. In recentyears, 2970 M. glyptostroboides logs ranging in diameter from 2 mto 8 m at the base have been discovered in the rice paddies (Yanget al., 2004; Cheng et al., 2008), indicating that the Shuisha valleywas covered by the forest before humans settled there.
4.3. Management and conservation implications
Since M. glyptostroboides was discovered in the 1940s, the Chi-nese government has made great efforts to protect the living fossil.However, only the adult trees of this species have received protec-tion, as the habitats and the associated species have been ignored(Wang et al., 2005). Official policy allows seeds to be collectedyearly from every adult tree, for sale by local villagers. If a fewseedlings were found in the stands, they were collected and movedto non-native habitats as landscape plants. Cultivation of profitableplants including C. fortunei, N. affinis, P. heteroclada, P. sulphurea, T.fortunei and C. chinensis in the stands containing M. glyptostroboideshas been common. The populations in the three areas show a dis-tinctive lack of young trees. Clearly, remedial action is needed. Theresults of this study will aid identification of priority areas for con-servation and forest management and restoration. Other threa-tened plants may also benefit: in some of the fragments inHabitat A1 we found Taxus chinensis var. mairei, and Cephalotaxusfortunei, important endangered species (Fu and Jin, 1992).
It may take over a 100 years to lose this long-lived species in itsnatural habitat, since adult plants are often less sensitive to chang-ing environmental conditions than seedlings and juveniles. Butthere is no guarantee of its continued existence there; effectivereproduction and recruitment will require strict habitat protectionfor Habitat A1. We urge that the collaboration of villagers be en-listed in efforts to allow reestablishment of the forests, and thatcontrols be imposed on the sale and transport of the seeds andseedlings of M. glyptostroboides. We suggest the creation of perma-nent plots in Habitat A1, and long-term monitoring on the regen-eration of the species.
5. Conclusion
The mixed habitats of M. glyptostrobioides have changed withinthe past 60 years. Habitat changes have eliminated seedlingrecruitment of the living fossil, and reduced species richness anddiversity in the community. Population structure analysis revealsno recruitment during the past 41 years. Human activity has beenthe major disturbing factor, and has included planting of otherprofitable species in the understory, selective cutting, harvestingfor fuel wood, and seed collection which began after the discoveryof the living species and its widespread propagation around theworld as an ornamental tree. The sites in Habitat A1 must have pri-ority in efforts leading toward recovery and conservation.
Acknowledgements
This study was financially supported by Natural Science Foun-dation of China 30700094. We are grateful to Kun Song of the EastChina Normal University and Ke Yang of the Chongqing Universityfor field assistance.
Appendix A. Supplementary material
Supplementary data associated with this article can be found, inthe online version, at doi:10.1016/j.biocon.2010.09.003.
References
Cavers, P.B., Harper, J.L., 1967. Studies in the dynamics of plant populations. I. Thefate of seed and transplants introduced into various habitats. J. Ecol. 55,59–71.
Chaney, R.W., 1948. The bearing of the living Metasequoia on problems of tertiarypaleobotany. Proc. Natl. Acad. USA 34 (11), 503–515.
Chaney, R.W., 1951. A revision of fossil Sequoia and Taxodium in western NorthAmerica based on the recent discovery of Metasequoia. Trans. Am. Philos. Soc. II40 (3), 171–263.
Chen, X.-Y., Li, Y.-Y., Wu, T.-Y., Zhang, X., Lu, H.-P., 2003. Size-class differences ingenetic structure of Metasequoia glyptostroboides Hu et Cheng (Taxodiaceae)plantations in Shanghai. Silvae Genet. 52, 107–109.
Cheng, W.C., Chu, Z.X., 1949. Forest status of Metasequoia glyptostroboides inLichuan, Hubei. Science (China) 31 (3), 73–80 (in Chinese).
Cheng, S.-L., Sun, Y.-Y., Yu, Z.-W., Shi, W., Xiang, J., Cheng, S.-M., 2008. Distributionand conservation of the buried wood of Metasequoia glyptostroboides in XingdouNature Reserve. For. Pract. Technol. 9, 35–36 (in Chinese).
Chu, K.-L., Cooper, W.S., 1950. An ecological reconnaissance in the native home ofMetasequoia glyptostroboides. Ecology 31 (2), 260–278.
Clark, J.S., Bechage, B., Camill, P., Cleveland, B., Hiller RisLamber, J., Lichter, J.,Mclanchlam, J., Mohan, J., Wickhoff, P., 1999. Interpreting recruitmentlimitation in forests. Am. J. Bot. 86, 1–16.
Cox, C.B., Moor, P.D., 1993. Biogeography. Blackwell Scientific Publications, Oxford.Florin, R., 1963. The distribution of conifer and taxad genera in time and space. Acta
Horti. Bergiana 20 (4), 1–312.Fu, L.G., Jin, J.M. (Eds.), 1992. China Plant Red Data Book: Rare and Endangered
Plants. Science Press, Beijing. p. 736.Gressitt, J.L., 1953. The California Academy – Lingnan Dawn Redwood Expedition.
Proc. California Acad. Sci., Fourth Series 28, 25–58.Hamrick, J.L., Godt, M.J.W., 1996. Conservation genetics of endemic plant species.
In: Avise, J.C., Hamrick, J.L. (Eds.), Conservation Genetics: Case Histories fromNature. Chapman and Hall, New York, pp. 281–304.
Hu, H.H., 1946. Notes on a Palaeogene species of Metasequoia in China. Bull. Geol.Soc. China 26, 105–107 (in Chinese).
Hu, H.H., 1948. How Metasequoia, the ‘‘living fossil” was discovered in China. J. NewYork Bot. Gard. 49, 201–202 (Excerpted in Arnoldia (1999) 59, 4–7).
Hu, H.H., Cheng, W.C., 1948. On the new family Metasequoiaceae and on Metasequoiaglyptostroboides, a living species of the genus Metasequoia found in Szechuanand Hupeh. Bull. Fan. Mem. Inst. Biol. New Ser. 1 (2), 153–161.
Iida, S., Nakashixuka, T., 1995. Forest fragmentation and its effect on speciesdiversity in sub-urban coppice forests in Japan. For. Ecol. Manag. 73,197–210.
IUCN, 2006. IUCN 2006 Red List of Threatened Species. www.iucnredlist.org.Karron, J.D., 1991. Patterns of genetic variation and breeding systems in rare plant
species. In: Falk, D.A., Holsinger, K.E. (Eds.), Genetics and Conservation of RarePlants. Oxford Univ. Press, New York, pp. 87–98.
Kikvidze, Z., Ohsawa, M., 2002. Measuring the number of co-dominants inecological communities. Ecol. Res. 17, 519–525.
Kuser, J.E., Sheely, D.L., Hendricks, D.R., 1997. Genetic variation in two ex situcollections of the rare Metasequoia glyptostroboides (Cupressaceae). SilvaeGenet. 46, 258–264.
Lande, R., 1988. Genetics and demography in biological conservation. Science 241,1455–1460.
Lande, R., 1998. Anthropogenic, ecological and genetic factors in extinction andconservation. Res. Popul. Ecol. (Kyoto) 40, 259–269.
Leng, Q., Fan, S.H., Li, W., Yang, H., Lai, X.L., Cheng, D.D., Ge, J.W., Shi, G.L., Jiang, Q.,Liu, X.Q., 2007. Database of native Metasequoia glyptostroboides trees in Chinabased on new census surveys and expeditions. Bull. Peabody Museum Nat. Hist.48 (2), 185–233.
LePage, B.A., Yang, H., Matsumoto, M., 2005. The evolution and biogeographichistory of Metasequoia. In: LePage, B.A., Williams, C.J., Yang, H. (Eds.), TheGeobiology and Ecology of Metasequoia. Springer, Dordrecht, pp. 3–114.
Li, Y.Y., 2006. Phylogenetic position and recovery assessment of Metasequoiaglyptostroboides. Dissertation. East China Normal Univ., Shanghai (in Chinese).
Li, J.H., Ban, J.D., 1989. The water fir communities endemic to China. J. HenanNormal Univ. (Natural Science Version) 4, 49–55 (in Chinese with Englishabstract).
288 C.Q. Tang et al. / Biological Conservation 144 (2011) 279–289
Author's personal copy
Li, Y.Y., Chen, X.Y., Zhang, X., Wu, T.Y., Lu, H.P., Cai, Y.W., 2005. Genetic differencesbetween wild and artificial populations of Metasequoia glyptostroboides:implications for species recovery. Conserv. Biol. 19, 224–231.
Magurran, A.E., 1988. Ecological Diversity and Its Measurement. Princeton Univ.Press.
MBL, 1958–2000. Meteorological Bureau of Lichuan, 1958–2000.Merrill, E.D., 1948. A living Metasequoia in China. Science 107, 140.Miki, S., 1941. On the change of flora in eastern Asia since Tertiary period (I). The
clay or lignite beds in Japan with special reference to the Pinus trifolia beds inthe central Hondo. Jpn. J. Bot. 11, 237–303.
Momohara, A., 1994. Floral and paleoenvironmental history from the late Plioceneto middle Pleistocene in and around central Japan. Palaeogeogr. Palaeoclimatol.Palaeoecol. 108, 281–293.
Momohara, A., 2005. Paleoecology and history of Metasequoia in Japan, withreferences to its extinction and survival in East Asia. In: LePage, B.A., Williams,C.J., Yang, H. (Eds.), The Geobiology and Ecology of Metasequoia. Springer,Dordrecht, pp. 115–136.
Myers, N., Mittermeier, R.A., Mittermeier, C.G., Fonseca, G.A.B., Kent, J., 2000.Biodiversity hotspots for conservation priorities. Nature 403, 853–858.
Ohsawa, M., 1984. Differentiation of vegetation zones and species strategies in thesubalpine region of Mt. Fuji. Vegetatio 57, 15–52.
Pielou, E.C., 1969. An Introduction to Mathematical Ecology. Wiley, New York.Schlarbaum, S.E., Tsuchiyu, T., 1984. Cytotaxonomy and phylogeny in certain
species of Taxodiaceae. Plant Syst. Evol. 147, 29–54.Schupp, E.W., Fuentes, M., 1995. Spatial patterns of seed dispersal and the
unification of plant population biology. Ecoscience 2, 267–275.Tamaki, I., Setsuko, S., Tomaru, N., 2008. Genetic variation and differentiation in
populations of a threatened tree, Magnolia stellata: factors influencing the levelof within-population genetic variation. Heredity 100, 415–423.
Tilman, D., May, R.M., Lehman, C.L., Nowak, M.A., 1994. Habitat destruction andextinction debt. Nature 371, 65–66.
Wang, X.Q., Ma, L.Y., Guo, B.X., Fan, S.H., Tan, J.X., 2005. Analysis on the changes ofthe original Metasequoia glyptostroboides population and its environment inLichuan, Hubei from 1948 to 2003. Acta Ecol. Sin. 25 (5), 972–977 (in Chinesewith English abstract).
Williams, C.B., 1947. The logarithmic series and its application to biologicalproblems. J. Ecol. 34, 253–273.
Williams, C.J., 2005. Ecological characteristics of Metasequoia glyptostroboides. In:LePage, B.A., Williams, C.J., Yang, H. (Eds.), The Geobiology and Ecology ofMetasequoia. Springer, Dordrecht, pp. 285–304.
Williams, C.J., LePage, B.A., Vann, D.R., Tange, T., Ikeda, H., Ando, M., Kusakabe,T., Tsuzuki, T., Sweda, T., 2003. Structure, allometry, and biomass ofplantation Metasequoia glyptostroboides in Japan. For. Ecol. Manag. 180(103), 287–301.
Wu, Z. (Ed.), 1980. Vegetation of China. Science Press, Beijing (in Chinese).Yang, H., 1999. From fossil to molecules: the Metasequoia tale continues. Arnoldia
58 (59), 60–71.Yang, H., 2005. Biomolecules from living and fossil Metasequoia: biological and
geological applications. In: LePage, B.A., Williams, C.J., Yang, H. (Eds.), TheGeobiology and Ecology of Metasequoia. Springer, Dordrecht, pp. 253–279.
Yang, H., Jin, J.H., 2000. Phytogeographic history and evolutionary stasis ofMetasequoia: geological and genetic information contrasted. Acta Palaeontol.Sin. 39 (Suppl.), 288–307.
Yang, J.M., Yang, X.Y., Liang, H., 2004. The discovery of buried Metasequoia wood inLichuan, Hubei, China, and its significance. Acta Palaeontol. Sin. 43 (1), 124–131(in Chinese).
You, D., Wang, Z., Lei, Y., Ma, G., 2008. Community classification and dynamicssuccession of natural forest of Metasequoia glyptostroboides Hu et Cheng. HubeiFor. Sci. Technol. 153, 6–11 (in Chinese with English abstract).
Yu, Y.H., 1995. Origin, evolution and distribution of the Taxodiaceae. Acta Phytotax.Sin. 33, 362–389.
C.Q. Tang et al. / Biological Conservation 144 (2011) 279–289 289